Input interpretation
potassium tetracyanonickelate(II) | nickel
Chemical names and formulas

| potassium tetracyanonickelate(II) | nickel formula | K_2Ni(CN)_4 | Ni Hill formula | C_4K_2N_4Ni-4 | Ni name | potassium tetracyanonickelate(II) | nickel IUPAC name | nickel; potassium; tetracyanide | nickel alternate names | nickel; potassium; tetracyanide | potassium nickel(II) cyanide | nickel metal mass fractions | C (carbon) 19.9% | K (potassium) 32.5% | N (nitrogen) 23.3% | Ni (nickel) 24.4% | Ni (nickel) 100%
Structure diagrams
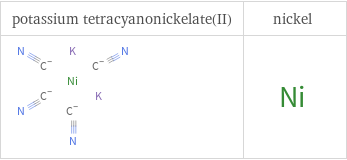
Structure diagrams
Basic properties

| potassium tetracyanonickelate(II) | nickel molar mass | 240.96 g/mol | 58.6934 g/mol phase | | solid (at STP) melting point | | 1453 °C boiling point | | 2732 °C density | | 8.908 g/cm^3 solubility in water | | insoluble
Units

Thermodynamic properties
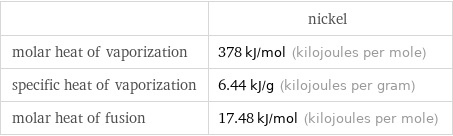
| nickel molar heat of vaporization | 378 kJ/mol (kilojoules per mole) specific heat of vaporization | 6.44 kJ/g (kilojoules per gram) molar heat of fusion | 17.48 kJ/mol (kilojoules per mole)
Solid properties
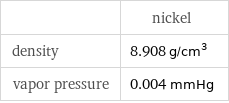
| nickel density | 8.908 g/cm^3 vapor pressure | 0.004 mmHg
Units

Chemical identifiers
![| potassium tetracyanonickelate(II) | nickel CAS number | 14220-17-8 | 7440-02-0 PubChem CID number | 6364988 | 935 PubChem SID number | 24878804 | 24852076 SMILES identifier | [C-]#N.[C-]#N.[C-]#N.[C-]#N.[K].[K].[Ni] | [Ni] InChI identifier | InChI=1/4CN.2K.Ni/c4*1-2;;;/q4*-1;;; | InChI=1/Ni RTECS number | QR8650000 | QR5950000 MDL number | MFCD00049669 | MFCD00011137](../image_source/8c8f66fed70abf571fb9317623750174.png)
| potassium tetracyanonickelate(II) | nickel CAS number | 14220-17-8 | 7440-02-0 PubChem CID number | 6364988 | 935 PubChem SID number | 24878804 | 24852076 SMILES identifier | [C-]#N.[C-]#N.[C-]#N.[C-]#N.[K].[K].[Ni] | [Ni] InChI identifier | InChI=1/4CN.2K.Ni/c4*1-2;;;/q4*-1;;; | InChI=1/Ni RTECS number | QR8650000 | QR5950000 MDL number | MFCD00049669 | MFCD00011137