Input interpretation

homonuclear diatomic molecules | electron affinity
Summary
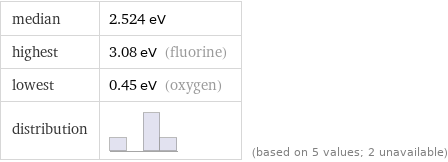
median | 2.524 eV highest | 3.08 eV (fluorine) lowest | 0.45 eV (oxygen) distribution | | (based on 5 values; 2 unavailable)
Entities with missing values
hydrogen | nitrogen (total: 2)
Ranked values
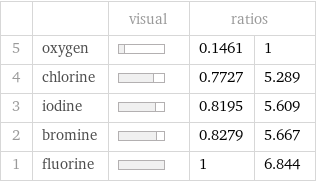
| | visual | ratios | 5 | oxygen | | 0.1461 | 1 4 | chlorine | | 0.7727 | 5.289 3 | iodine | | 0.8195 | 5.609 2 | bromine | | 0.8279 | 5.667 1 | fluorine | | 1 | 6.844
Electron affinity rankings

1 | oxygen | 0.45 eV 2 | chlorine | 2.38 eV 3 | iodine | 2.524 eV 4 | bromine | 2.55 eV 5 | fluorine | 3.08 eV (based on 5 values; 2 unavailable)