Input interpretation
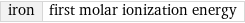
iron | first molar ionization energy
Result
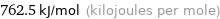
762.5 kJ/mol (kilojoules per mole)
Unit conversions
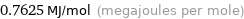
0.7625 MJ/mol (megajoules per mole)
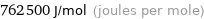
762500 J/mol (joules per mole)
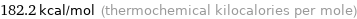
182.2 kcal/mol (thermochemical kilocalories per mole)

7.903 eV (molar electronvolts)
Comparisons as molar ionization energy

≈ 0.32 × molar energy required to remove the first electron from helium ( 2372 kJ/mol )
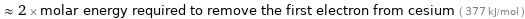
≈ 2 × molar energy required to remove the first electron from cesium ( 377 kJ/mol )