Input interpretation
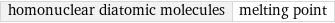
homonuclear diatomic molecules | melting point
Summary

median | -210 °C highest | 113 °C (iodine) lowest | -259.2 °C (hydrogen) distribution |
Melting point rankings

1 | hydrogen | -259.2 °C 2 | fluorine | -219.6 °C 3 | oxygen | -218 °C 4 | nitrogen | -210 °C 5 | chlorine | -101 °C 6 | bromine | -7.2 °C 7 | iodine | 113 °C
Unit conversions for median melting point -210 °C
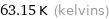
63.15 K (kelvins)

-346 °F (degrees Fahrenheit)
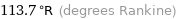
113.7 °R (degrees Rankine)
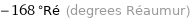
-168 °Ré (degrees Réaumur)

-102.8 °Rø (degrees Rømer)
Comparison for median melting point -210 °C
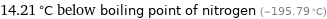
14.21 °C below boiling point of nitrogen (-195.79 °C)
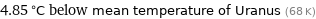
4.85 °C below mean temperature of Uranus (68 K)
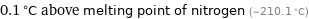
0.1 °C above melting point of nitrogen (-210.1 °C)
Corresponding quantities

Thermodynamic energy E from E = kT: | 5.4 meV (millielectronvolts)
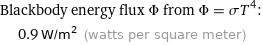
Blackbody energy flux Φ from Φ = σT^4: | 0.9 W/m^2 (watts per square meter)