Input interpretation
alkaline earth metal ions
Members
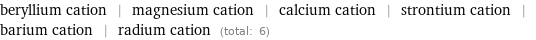
beryllium cation | magnesium cation | calcium cation | strontium cation | barium cation | radium cation (total: 6)
Structure diagrams
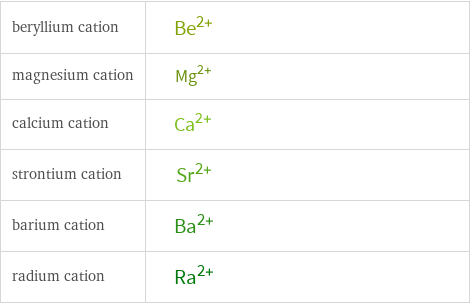
Structure diagrams
Ionic radii
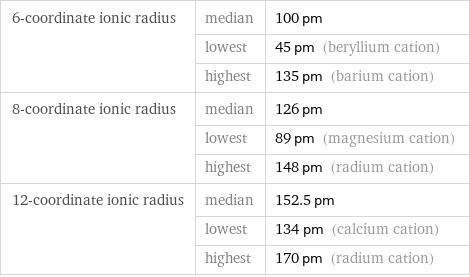
6-coordinate ionic radius | median | 100 pm | lowest | 45 pm (beryllium cation) | highest | 135 pm (barium cation) 8-coordinate ionic radius | median | 126 pm | lowest | 89 pm (magnesium cation) | highest | 148 pm (radium cation) 12-coordinate ionic radius | median | 152.5 pm | lowest | 134 pm (calcium cation) | highest | 170 pm (radium cation)
Units
