Input interpretation

alkali metals | critical temperature
Summary
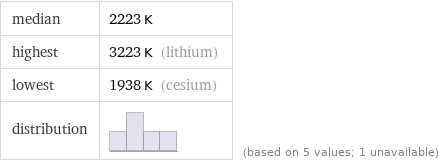
median | 2223 K highest | 3223 K (lithium) lowest | 1938 K (cesium) distribution | | (based on 5 values; 1 unavailable)
Entity with missing value
francium
Ranked values
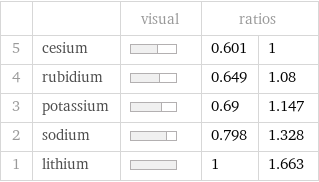
| | visual | ratios | 5 | cesium | | 0.601 | 1 4 | rubidium | | 0.649 | 1.08 3 | potassium | | 0.69 | 1.147 2 | sodium | | 0.798 | 1.328 1 | lithium | | 1 | 1.663
Critical temperature rankings
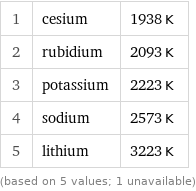
1 | cesium | 1938 K 2 | rubidium | 2093 K 3 | potassium | 2223 K 4 | sodium | 2573 K 5 | lithium | 3223 K (based on 5 values; 1 unavailable)
Unit conversions for median critical temperature 2223 K
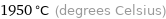
1950 °C (degrees Celsius)

3542 °F (degrees Fahrenheit)
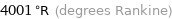
4001 °R (degrees Rankine)

1560 °Ré (degrees Réaumur)
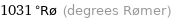
1031 °Rø (degrees Rømer)
Comparison for median critical temperature 2223 K
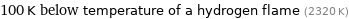
100 K below temperature of a hydrogen flame (2320 K)
Corresponding quantities
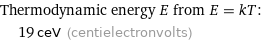
Thermodynamic energy E from E = kT: | 19 ceV (centielectronvolts)
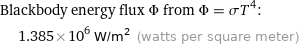
Blackbody energy flux Φ from Φ = σT^4: | 1.385×10^6 W/m^2 (watts per square meter)

Approximate luminous exitance from a planar blackbody radiator perpendicular to its surface: | 5.074×10^6 lx (lux)