Input interpretation
sodium bromate | lithium bromide
Chemical names and formulas
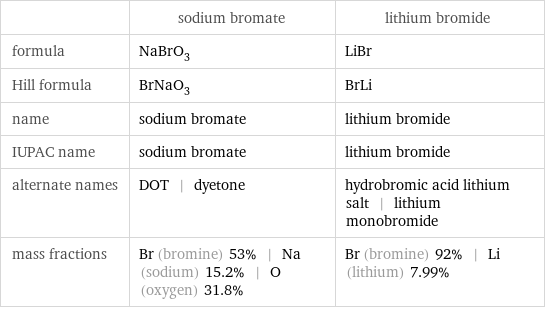
| sodium bromate | lithium bromide formula | NaBrO_3 | LiBr Hill formula | BrNaO_3 | BrLi name | sodium bromate | lithium bromide IUPAC name | sodium bromate | lithium bromide alternate names | DOT | dyetone | hydrobromic acid lithium salt | lithium monobromide mass fractions | Br (bromine) 53% | Na (sodium) 15.2% | O (oxygen) 31.8% | Br (bromine) 92% | Li (lithium) 7.99%
Structure diagrams
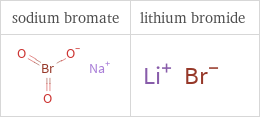
Structure diagrams
Basic properties
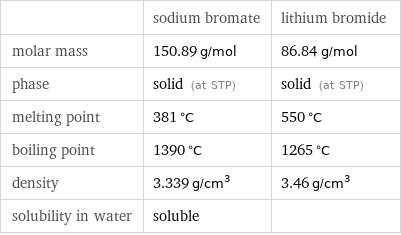
| sodium bromate | lithium bromide molar mass | 150.89 g/mol | 86.84 g/mol phase | solid (at STP) | solid (at STP) melting point | 381 °C | 550 °C boiling point | 1390 °C | 1265 °C density | 3.339 g/cm^3 | 3.46 g/cm^3 solubility in water | soluble |
Units

Thermodynamic properties
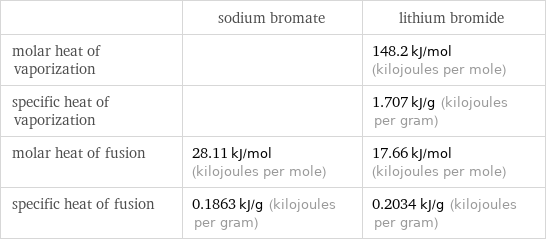
| sodium bromate | lithium bromide molar heat of vaporization | | 148.2 kJ/mol (kilojoules per mole) specific heat of vaporization | | 1.707 kJ/g (kilojoules per gram) molar heat of fusion | 28.11 kJ/mol (kilojoules per mole) | 17.66 kJ/mol (kilojoules per mole) specific heat of fusion | 0.1863 kJ/g (kilojoules per gram) | 0.2034 kJ/g (kilojoules per gram)
Solid properties (at STP)

| sodium bromate | lithium bromide density | 3.339 g/cm^3 | 3.46 g/cm^3 vapor pressure | | 50 mmHg refractive index | | 1.784
Units
