Input interpretation

methane | formal charges
Result
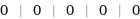
0 | 0 | 0 | 0 | 0
Lewis structure

Draw the Lewis structure of methane. Start by drawing the overall structure of the molecule: Count the total valence electrons of the carbon (n_C, val = 4) and hydrogen (n_H, val = 1) atoms: n_C, val + 4 n_H, val = 8 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8) and hydrogen (n_H, full = 2): n_C, full + 4 n_H, full = 16 Subtracting these two numbers shows that 16 - 8 = 8 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 4 bonds and hence 8 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 8 - 8 = 0 electrons left to draw and the diagram is complete: Answer: | |