Input interpretation
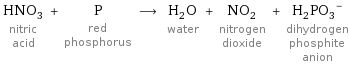
HNO_3 nitric acid + P red phosphorus ⟶ H_2O water + NO_2 nitrogen dioxide + (H_2PO_3)^- dihydrogen phosphite anion
Chemical names and formulas

| nitric acid | red phosphorus | water | nitrogen dioxide | dihydrogen phosphite anion formula | HNO_3 | P | H_2O | NO_2 | (H_2PO_3)^- name | nitric acid | red phosphorus | water | nitrogen dioxide | dihydrogen phosphite anion IUPAC name | nitric acid | phosphorus | water | Nitrogen dioxide |