Input interpretation
copper hydroxide | rubidium fluoride
Chemical names and formulas
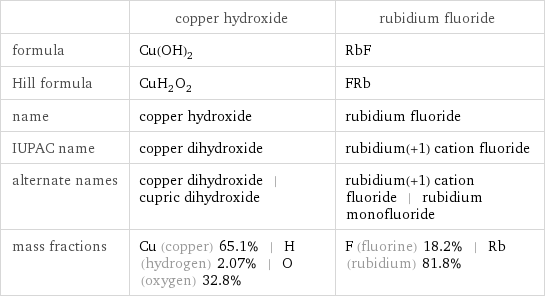
| copper hydroxide | rubidium fluoride formula | Cu(OH)_2 | RbF Hill formula | CuH_2O_2 | FRb name | copper hydroxide | rubidium fluoride IUPAC name | copper dihydroxide | rubidium(+1) cation fluoride alternate names | copper dihydroxide | cupric dihydroxide | rubidium(+1) cation fluoride | rubidium monofluoride mass fractions | Cu (copper) 65.1% | H (hydrogen) 2.07% | O (oxygen) 32.8% | F (fluorine) 18.2% | Rb (rubidium) 81.8%
Structure diagrams
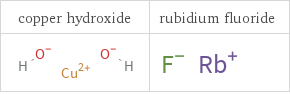
Structure diagrams
Basic properties
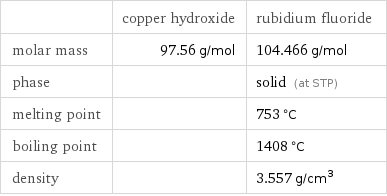
| copper hydroxide | rubidium fluoride molar mass | 97.56 g/mol | 104.466 g/mol phase | | solid (at STP) melting point | | 753 °C boiling point | | 1408 °C density | | 3.557 g/cm^3
Units
