Input interpretation
boron trifluoride
Chemical names and formulas

formula | BF_3 name | boron trifluoride IUPAC name | trifluoroborane alternate names | borane, trifluoro- | boron fluoride | boron fluoride (1:3) | trifluoroborane mass fractions | B (boron) 15.9% | F (fluorine) 84.1%
Lewis structure

Draw the Lewis structure of boron trifluoride. Start by drawing the overall structure of the molecule: Count the total valence electrons of the boron (n_B, val = 3) and fluorine (n_F, val = 7) atoms: n_B, val + 3 n_F, val = 24 Calculate the number of electrons needed to completely fill the valence shells for boron (n_B, full = 6) and fluorine (n_F, full = 8): n_B, full + 3 n_F, full = 30 Subtracting these two numbers shows that 30 - 24 = 6 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 3 bonds and hence 6 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 24 - 6 = 18 electrons left to draw: Answer: | |
Basic properties

molar mass | 67.81 g/mol phase | gas (at STP) melting point | -127 °C boiling point | -100 °C density | 0.002772 g/cm^3 (at 25 °C) dielectric constant | 1.001
Gas properties (at STP)
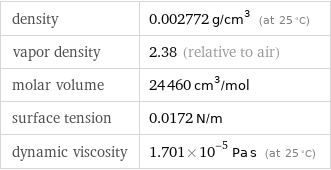
density | 0.002772 g/cm^3 (at 25 °C) vapor density | 2.38 (relative to air) molar volume | 24460 cm^3/mol surface tension | 0.0172 N/m dynamic viscosity | 1.701×10^-5 Pa s (at 25 °C)
Units

Thermodynamic properties

specific free energy of formation Δ_fG° | gas | -16.51 kJ/g molar free energy of formation Δ_fG° | gas | -1119 kJ/mol specific heat of formation Δ_fH° | gas | -16.75 kJ/g molar heat of formation Δ_fH° | gas | -1136 kJ/mol molar heat of vaporization | 17 kJ/mol | specific heat of vaporization | 0.251 kJ/g | molar heat of fusion | 4.2 kJ/mol | specific heat of fusion | 0.062 kJ/g | critical temperature | 260.9 K | critical pressure | 4.99 MPa | (at STP)
Chemical identifiers

CAS number | 7637-07-2 PubChem CID number | 6356 PubChem SID number | 24857753 SMILES identifier | B(F)(F)F InChI identifier | InChI=1/BF3/c2-1(3)4 RTECS number | ED2275000 MDL number | MFCD00011316
NFPA label

NFPA label

NFPA health rating | 4 NFPA fire rating | 0 NFPA reactivity rating | 1
Safety properties

flash point | 4 °C

DOT hazard class | 2.3 DOT numbers | 1008