Input information

compressibility factor for van der Waals gas | temperature | 30 °C (degrees Celsius) critical temperature | water (critical temperature): 647.14 K (kelvins) pressure | 200 atm (atmospheres) critical pressure | 37.9 atm (atmospheres)
Results
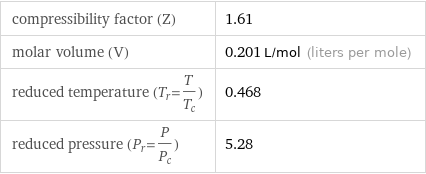
compressibility factor (Z) | 1.61 molar volume (V) | 0.201 L/mol (liters per mole) reduced temperature (T_r=T/T_c) | 0.468 reduced pressure (P_r=P/P_c) | 5.28
Compressibility factor plot
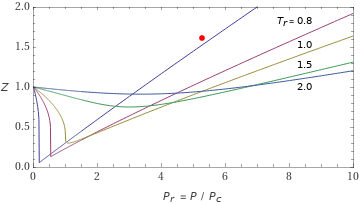
Compressibility factor plot