Input interpretation
carbon monoxide
Chemical names and formulas

formula | CO name | carbon monoxide alternate names | carbonic oxide | carbon monooxide | carbon oxide | monoxide mass fractions | C (carbon) 42.9% | O (oxygen) 57.1%
Lewis structure

Draw the Lewis structure of carbon monoxide. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the carbon (n_C, val = 4) and oxygen (n_O, val = 6) atoms: n_C, val + n_O, val = 10 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8) and oxygen (n_O, full = 8): n_C, full + n_O, full = 16 Subtracting these two numbers shows that 16 - 10 = 6 bonding electrons are needed. Each bond has two electrons, so in addition to the 1 bond already present in the diagram add 2 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: Fill in the 2 bonds by pairing electrons between adjacent highlighted atoms, noting the formal charges of the atoms. The triple bond is a dipolar bond with oxygen donating both electrons: Answer: | |
3D structure

3D structure
Basic properties
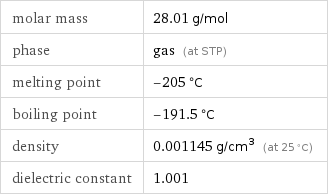
molar mass | 28.01 g/mol phase | gas (at STP) melting point | -205 °C boiling point | -191.5 °C density | 0.001145 g/cm^3 (at 25 °C) dielectric constant | 1.001
Gas properties (at STP)
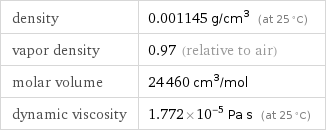
density | 0.001145 g/cm^3 (at 25 °C) vapor density | 0.97 (relative to air) molar volume | 24460 cm^3/mol dynamic viscosity | 1.772×10^-5 Pa s (at 25 °C)
Units
