Input interpretation
sodium bromate | cobaltous nitrate hexahydrate
Chemical names and formulas
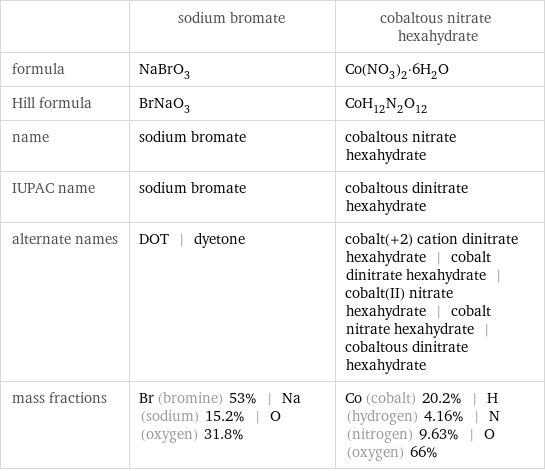
| sodium bromate | cobaltous nitrate hexahydrate formula | NaBrO_3 | Co(NO_3)_2·6H_2O Hill formula | BrNaO_3 | CoH_12N_2O_12 name | sodium bromate | cobaltous nitrate hexahydrate IUPAC name | sodium bromate | cobaltous dinitrate hexahydrate alternate names | DOT | dyetone | cobalt(+2) cation dinitrate hexahydrate | cobalt dinitrate hexahydrate | cobalt(II) nitrate hexahydrate | cobalt nitrate hexahydrate | cobaltous dinitrate hexahydrate mass fractions | Br (bromine) 53% | Na (sodium) 15.2% | O (oxygen) 31.8% | Co (cobalt) 20.2% | H (hydrogen) 4.16% | N (nitrogen) 9.63% | O (oxygen) 66%
Structure diagrams
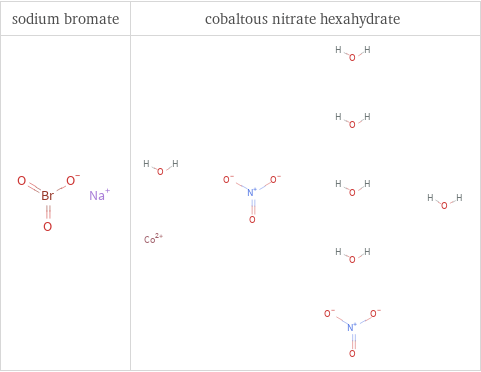
Structure diagrams
Basic properties
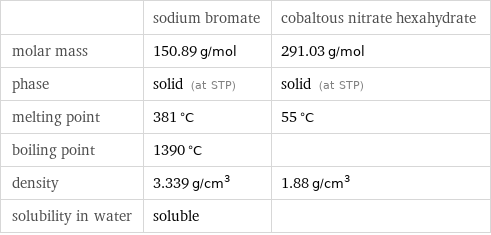
| sodium bromate | cobaltous nitrate hexahydrate molar mass | 150.89 g/mol | 291.03 g/mol phase | solid (at STP) | solid (at STP) melting point | 381 °C | 55 °C boiling point | 1390 °C | density | 3.339 g/cm^3 | 1.88 g/cm^3 solubility in water | soluble |
Units

Thermodynamic properties
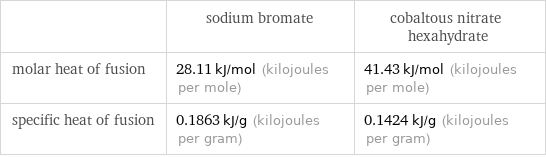
| sodium bromate | cobaltous nitrate hexahydrate molar heat of fusion | 28.11 kJ/mol (kilojoules per mole) | 41.43 kJ/mol (kilojoules per mole) specific heat of fusion | 0.1863 kJ/g (kilojoules per gram) | 0.1424 kJ/g (kilojoules per gram)
Solid properties (at STP)
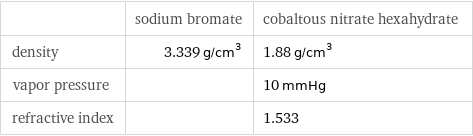
| sodium bromate | cobaltous nitrate hexahydrate density | 3.339 g/cm^3 | 1.88 g/cm^3 vapor pressure | | 10 mmHg refractive index | | 1.533
Units

Chemical identifiers
![| sodium bromate | cobaltous nitrate hexahydrate CAS number | 7789-38-0 | 10026-22-9 PubChem CID number | 23668195 | 24821 PubChem SID number | 24853461 | 24852146 SMILES identifier | [O-]Br(=O)=O.[Na+] | [N+](=O)([O-])[O-].[N+](=O)([O-])[O-].O.O.O.O.O.O.[Co+2]](../image_source/008823d64a0749397dc4a4dfd1f402d6.png)
| sodium bromate | cobaltous nitrate hexahydrate CAS number | 7789-38-0 | 10026-22-9 PubChem CID number | 23668195 | 24821 PubChem SID number | 24853461 | 24852146 SMILES identifier | [O-]Br(=O)=O.[Na+] | [N+](=O)([O-])[O-].[N+](=O)([O-])[O-].O.O.O.O.O.O.[Co+2]