Input interpretation
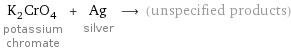
K_2CrO_4 potassium chromate + Ag silver ⟶ (unspecified products)
Chemical names and formulas

| potassium chromate | silver formula | K_2CrO_4 | Ag Hill formula | CrK_2O_4 | Ag name | potassium chromate | silver IUPAC name | dipotassium dioxido-dioxochromium | silver
Substance properties
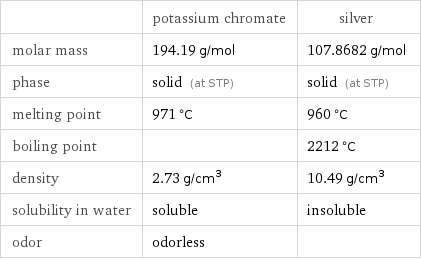
| potassium chromate | silver molar mass | 194.19 g/mol | 107.8682 g/mol phase | solid (at STP) | solid (at STP) melting point | 971 °C | 960 °C boiling point | | 2212 °C density | 2.73 g/cm^3 | 10.49 g/cm^3 solubility in water | soluble | insoluble odor | odorless |
Units
