Input interpretation
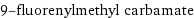
9-fluorenylmethyl carbamate
Basic properties
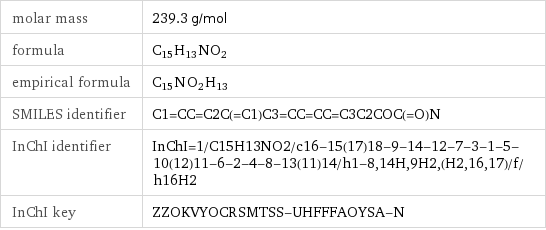
molar mass | 239.3 g/mol formula | C_15H_13NO_2 empirical formula | C_15N_O_2H_13 SMILES identifier | C1=CC=C2C(=C1)C3=CC=CC=C3C2COC(=O)N InChI identifier | InChI=1/C15H13NO2/c16-15(17)18-9-14-12-7-3-1-5-10(12)11-6-2-4-8-13(11)14/h1-8, 14H, 9H2, (H2, 16, 17)/f/h16H2 InChI key | ZZOKVYOCRSMTSS-UHFFFAOYSA-N
Lewis structure

Draw the Lewis structure of 9-fluorenylmethyl carbamate. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), nitrogen (n_N, val = 5), and oxygen (n_O, val = 6) atoms: 15 n_C, val + 13 n_H, val + n_N, val + 2 n_O, val = 90 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), nitrogen (n_N, full = 8), and oxygen (n_O, full = 8): 15 n_C, full + 13 n_H, full + n_N, full + 2 n_O, full = 170 Subtracting these two numbers shows that 170 - 90 = 80 bonding electrons are needed. Each bond has two electrons, so in addition to the 33 bonds already present in the diagram add 7 bonds. To minimize formal charge carbon wants 4 bonds and oxygen wants 2 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: Fill in the 7 bonds by pairing electrons between adjacent highlighted atoms. Note that the six atom rings are aromatic, so that the single and double bonds may be rearranged: Answer: | |
Estimated thermodynamic properties
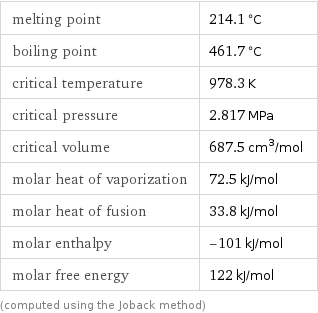
melting point | 214.1 °C boiling point | 461.7 °C critical temperature | 978.3 K critical pressure | 2.817 MPa critical volume | 687.5 cm^3/mol molar heat of vaporization | 72.5 kJ/mol molar heat of fusion | 33.8 kJ/mol molar enthalpy | -101 kJ/mol molar free energy | 122 kJ/mol (computed using the Joback method)
Units
Quantitative molecular descriptors
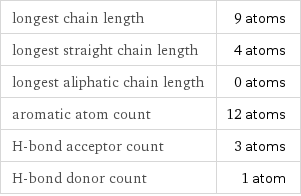
longest chain length | 9 atoms longest straight chain length | 4 atoms longest aliphatic chain length | 0 atoms aromatic atom count | 12 atoms H-bond acceptor count | 3 atoms H-bond donor count | 1 atom
Elemental composition

Find the elemental composition for 9-fluorenylmethyl carbamate in terms of the atom and mass percents: atom percent = N_i/N_atoms × 100% mass percent = (N_im_i)/m × 100% Plan: • Write the chemical formula and gather atomic masses from the periodic table. • Determine values for N_i, m_i, N_atoms and m using these items. • Finally, compute the percents and check the results. Write the chemical formula: C_15H_13NO_2 Use the chemical formula to count the number of atoms, N_i, for each element and find the total number of atoms, N_atoms, per molecule: | number of atoms C (carbon) | 15 N (nitrogen) | 1 O (oxygen) | 2 H (hydrogen) | 13 N_atoms = 15 + 1 + 2 + 13 = 31 Divide each N_i by N_atoms to calculate atom fractions. Then use the property that atom fractions must sum to one to check the work: | number of atoms | atom fraction C (carbon) | 15 | 15/31 N (nitrogen) | 1 | 1/31 O (oxygen) | 2 | 2/31 H (hydrogen) | 13 | 13/31 Check: 15/31 + 1/31 + 2/31 + 13/31 = 1 Compute atom percents using the atom fractions: | number of atoms | atom percent C (carbon) | 15 | 15/31 × 100% = 48.4% N (nitrogen) | 1 | 1/31 × 100% = 3.23% O (oxygen) | 2 | 2/31 × 100% = 6.45% H (hydrogen) | 13 | 13/31 × 100% = 41.9% Look up the atomic mass, m_i, in unified atomic mass units, u, for each element in the periodic table: | number of atoms | atom percent | atomic mass/u C (carbon) | 15 | 48.4% | 12.011 N (nitrogen) | 1 | 3.23% | 14.007 O (oxygen) | 2 | 6.45% | 15.999 H (hydrogen) | 13 | 41.9% | 1.008 Multiply N_i by m_i to compute the mass for each element. Then sum those values to compute the molecular mass, m: | number of atoms | atom percent | atomic mass/u | mass/u C (carbon) | 15 | 48.4% | 12.011 | 15 × 12.011 = 180.165 N (nitrogen) | 1 | 3.23% | 14.007 | 1 × 14.007 = 14.007 O (oxygen) | 2 | 6.45% | 15.999 | 2 × 15.999 = 31.998 H (hydrogen) | 13 | 41.9% | 1.008 | 13 × 1.008 = 13.104 m = 180.165 u + 14.007 u + 31.998 u + 13.104 u = 239.274 u Divide the mass for each element by m to calculate mass fractions. Then use the property that mass fractions must sum to one to check the work: | number of atoms | atom percent | mass fraction C (carbon) | 15 | 48.4% | 180.165/239.274 N (nitrogen) | 1 | 3.23% | 14.007/239.274 O (oxygen) | 2 | 6.45% | 31.998/239.274 H (hydrogen) | 13 | 41.9% | 13.104/239.274 Check: 180.165/239.274 + 14.007/239.274 + 31.998/239.274 + 13.104/239.274 = 1 Compute mass percents using the mass fractions: Answer: | | | number of atoms | atom percent | mass percent C (carbon) | 15 | 48.4% | 180.165/239.274 × 100% = 75.30% N (nitrogen) | 1 | 3.23% | 14.007/239.274 × 100% = 5.854% O (oxygen) | 2 | 6.45% | 31.998/239.274 × 100% = 13.37% H (hydrogen) | 13 | 41.9% | 13.104/239.274 × 100% = 5.477%
Elemental oxidation states
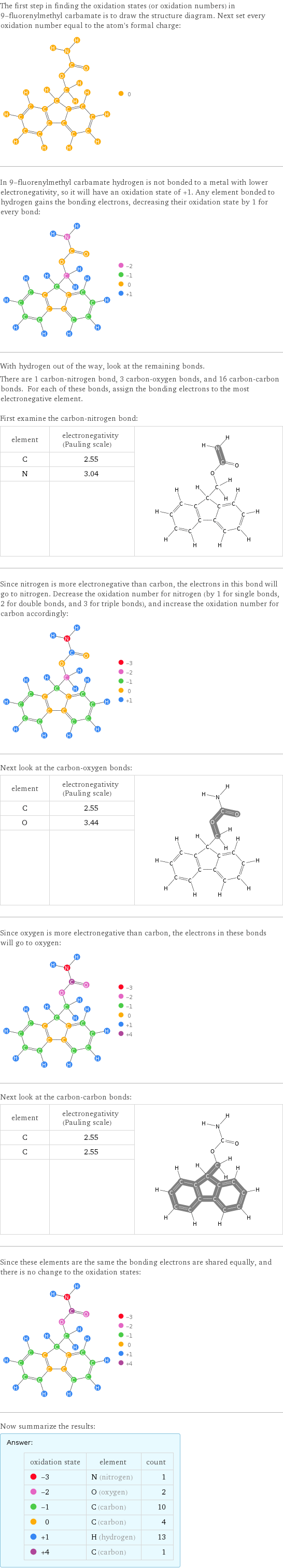
The first step in finding the oxidation states (or oxidation numbers) in 9-fluorenylmethyl carbamate is to draw the structure diagram. Next set every oxidation number equal to the atom's formal charge: In 9-fluorenylmethyl carbamate hydrogen is not bonded to a metal with lower electronegativity, so it will have an oxidation state of +1. Any element bonded to hydrogen gains the bonding electrons, decreasing their oxidation state by 1 for every bond: With hydrogen out of the way, look at the remaining bonds. There are 1 carbon-nitrogen bond, 3 carbon-oxygen bonds, and 16 carbon-carbon bonds. For each of these bonds, assign the bonding electrons to the most electronegative element. First examine the carbon-nitrogen bond: element | electronegativity (Pauling scale) | C | 2.55 | N | 3.04 | | | Since nitrogen is more electronegative than carbon, the electrons in this bond will go to nitrogen. Decrease the oxidation number for nitrogen (by 1 for single bonds, 2 for double bonds, and 3 for triple bonds), and increase the oxidation number for carbon accordingly: Next look at the carbon-oxygen bonds: element | electronegativity (Pauling scale) | C | 2.55 | O | 3.44 | | | Since oxygen is more electronegative than carbon, the electrons in these bonds will go to oxygen: Next look at the carbon-carbon bonds: element | electronegativity (Pauling scale) | C | 2.55 | C | 2.55 | | | Since these elements are the same the bonding electrons are shared equally, and there is no change to the oxidation states: Now summarize the results: Answer: | | oxidation state | element | count -3 | N (nitrogen) | 1 -2 | O (oxygen) | 2 -1 | C (carbon) | 10 0 | C (carbon) | 4 +1 | H (hydrogen) | 13 +4 | C (carbon) | 1
Orbital hybridization
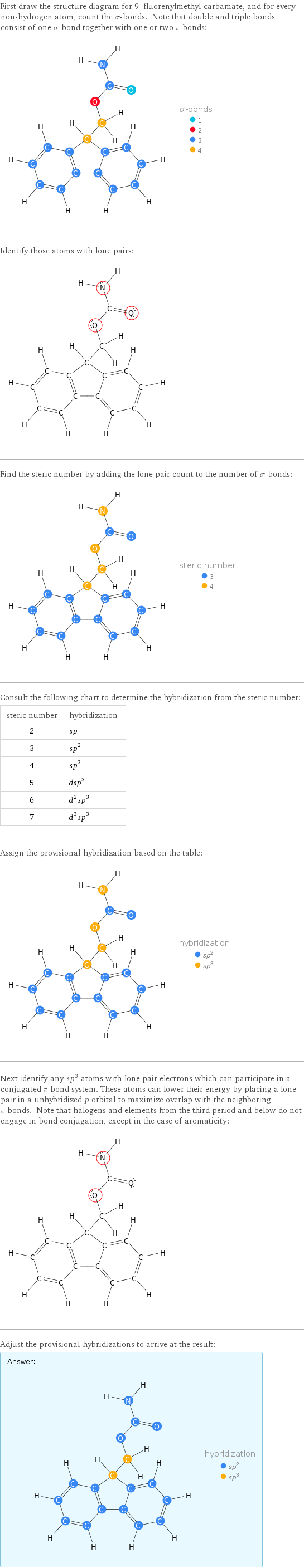
First draw the structure diagram for 9-fluorenylmethyl carbamate, and for every non-hydrogen atom, count the σ-bonds. Note that double and triple bonds consist of one σ-bond together with one or two π-bonds: Identify those atoms with lone pairs: Find the steric number by adding the lone pair count to the number of σ-bonds: Consult the following chart to determine the hybridization from the steric number: steric number | hybridization 2 | sp 3 | sp^2 4 | sp^3 5 | dsp^3 6 | d^2sp^3 7 | d^3sp^3 Assign the provisional hybridization based on the table: Next identify any sp^3 atoms with lone pair electrons which can participate in a conjugated π-bond system. These atoms can lower their energy by placing a lone pair in a unhybridized p orbital to maximize overlap with the neighboring π-bonds. Note that halogens and elements from the third period and below do not engage in bond conjugation, except in the case of aromaticity: Adjust the provisional hybridizations to arrive at the result: Answer: | |
Topological indices
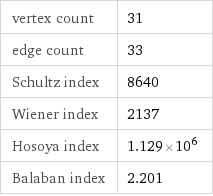
vertex count | 31 edge count | 33 Schultz index | 8640 Wiener index | 2137 Hosoya index | 1.129×10^6 Balaban index | 2.201