Input interpretation

potassium iodate | orbital hybridization
Result
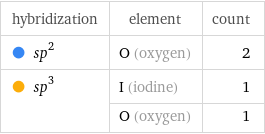
hybridization | element | count sp^2 | O (oxygen) | 2 sp^3 | I (iodine) | 1 | O (oxygen) | 1
Chemical names and formulas

formula | KIO_3 Hill formula | IKO_3 name | potassium iodate alternate names | iodic acid potassium salt | potassium iodine oxide mass fractions | I (iodine) 59.3% | K (potassium) 18.3% | O (oxygen) 22.4%
Ion equivalents
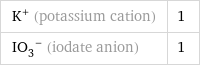
K^+ (potassium cation) | 1 (IO_3)^- (iodate anion) | 1