Input interpretation
chlorate anion | hydrogen borate anion | periodate anion
Structure diagrams
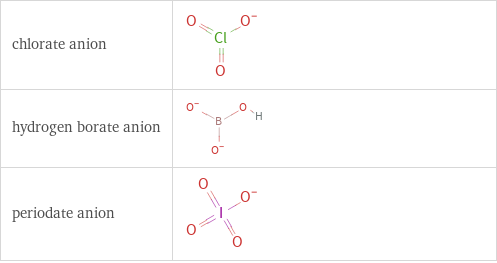
Structure diagrams
General properties
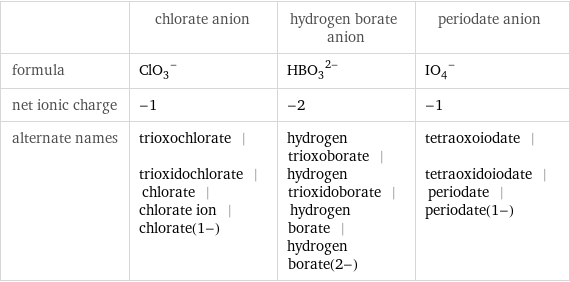
| chlorate anion | hydrogen borate anion | periodate anion formula | (ClO_3)^- | (HBO_3)^(2-) | (IO_4)^- net ionic charge | -1 | -2 | -1 alternate names | trioxochlorate | trioxidochlorate | chlorate | chlorate ion | chlorate(1-) | hydrogen trioxoborate | hydrogen trioxidoborate | hydrogen borate | hydrogen borate(2-) | tetraoxoiodate | tetraoxidoiodate | periodate | periodate(1-)
Ionic radii

| chlorate anion | periodate anion thermochemical radius | 171 pm | 249 pm
Units

Other properties
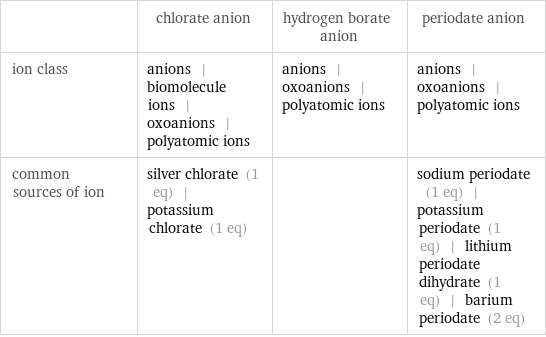
| chlorate anion | hydrogen borate anion | periodate anion ion class | anions | biomolecule ions | oxoanions | polyatomic ions | anions | oxoanions | polyatomic ions | anions | oxoanions | polyatomic ions common sources of ion | silver chlorate (1 eq) | potassium chlorate (1 eq) | | sodium periodate (1 eq) | potassium periodate (1 eq) | lithium periodate dihydrate (1 eq) | barium periodate (2 eq)