Input interpretation

period 5 elements | electrical conductivity
Summary

median | 8.3×10^6 S/m highest | 6.2×10^7 S/m (silver) lowest | 1×10^-7 S/m (iodine) distribution | | (based on 17 values; 1 unavailable)
Entity with missing value
xenon
Plot
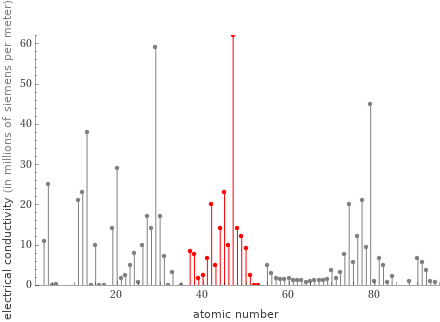
Plot
Periodic table location

Periodic table location
Distribution plots
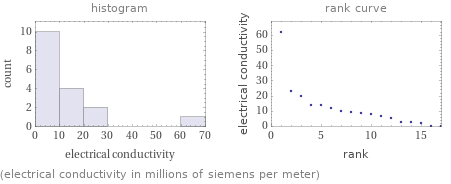
(electrical conductivity in millions of siemens per meter)
Electrical conductivity rankings
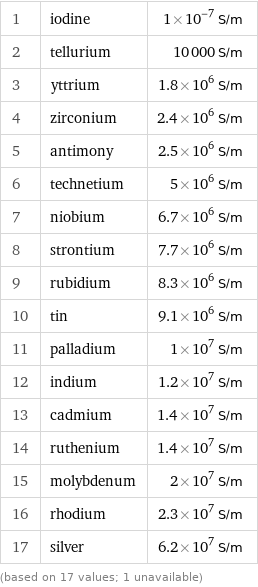
1 | iodine | 1×10^-7 S/m 2 | tellurium | 10000 S/m 3 | yttrium | 1.8×10^6 S/m 4 | zirconium | 2.4×10^6 S/m 5 | antimony | 2.5×10^6 S/m 6 | technetium | 5×10^6 S/m 7 | niobium | 6.7×10^6 S/m 8 | strontium | 7.7×10^6 S/m 9 | rubidium | 8.3×10^6 S/m 10 | tin | 9.1×10^6 S/m 11 | palladium | 1×10^7 S/m 12 | indium | 1.2×10^7 S/m 13 | cadmium | 1.4×10^7 S/m 14 | ruthenium | 1.4×10^7 S/m 15 | molybdenum | 2×10^7 S/m 16 | rhodium | 2.3×10^7 S/m 17 | silver | 6.2×10^7 S/m (based on 17 values; 1 unavailable)
Unit conversions for median electrical conductivity 8.3×10^6 S/m

8.3 MS/m (megasiemens per meter)

8300 S/mm (siemens per milimeter)
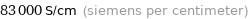
83000 S/cm (siemens per centimeter)

0.0083 ab℧/m (abmhos per meter) (unit officially deprecated)
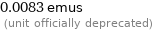
0.0083 emus (unit officially deprecated)

7.5×10^16 esus (unit officially deprecated)
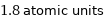
1.8 atomic units
Comparisons for median electrical conductivity 8.3×10^6 S/m
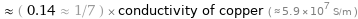
≈ ( 0.14 ≈ 1/7 ) × conductivity of copper ( ≈ 5.9×10^7 S/m )
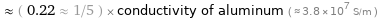
≈ ( 0.22 ≈ 1/5 ) × conductivity of aluminum ( ≈ 3.8×10^7 S/m )
Corresponding quantity
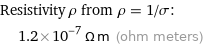
Resistivity ρ from ρ = 1/σ: | 1.2×10^-7 Ω m (ohm meters)