Input interpretation

cobalt(II) cation | ion class
Result
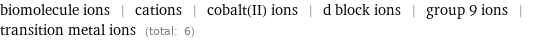
biomolecule ions | cations | cobalt(II) ions | d block ions | group 9 ions | transition metal ions (total: 6)
Ionic radii

| 2-coordinate | 4-coordinate biomolecule ions | 57.5 pm (median) 46 pm (copper(I) cation) (lowest) 69 pm (mercury(II) cation) (highest) | 60 pm (median) 26 pm (chromium(VI) cation) (lowest) 137 pm (potassium cation) (highest) cations | 57.5 pm (median) 46 pm (copper(I) cation) (lowest) 69 pm (mercury(II) cation) (highest) | 61.5 pm (median) 39 pm (aluminum cation) (lowest) 137 pm (potassium cation) (highest) cobalt(II) ions | | 56 pm (median) 56 pm (cobalt(II) cation) (lowest) 56 pm (cobalt(II) cation) (highest) d block ions | 57.5 pm (median) 46 pm (copper(I) cation) (lowest) 69 pm (mercury(II) cation) (highest) | 59.5 pm (median) 25 pm (manganese(VII) cation) (lowest) 100 pm (silver(I) cation) (highest) group 9 ions | | 56 pm (median) 56 pm (cobalt(II) cation) (lowest) 56 pm (cobalt(II) cation) (highest) transition metal ions | 69 pm (median) 69 pm (mercury(II) cation) (lowest) 69 pm (mercury(II) cation) (highest) | 60 pm (median) 36 pm (vanadium(V) cation) (lowest) 100 pm (silver(I) cation) (highest) | 5-coordinate | 6-coordinate biomolecule ions | 49.5 pm (median) 48 pm (aluminum cation) (lowest) 51 pm (tungsten(VI) cation) (highest) | 84.5 pm (median) 44 pm (chromium(VI) cation) (lowest) 220 pm (iodide anion) (highest) cations | 48 pm (median) 48 pm (aluminum cation) (lowest) 48 pm (aluminum cation) (highest) | 73 pm (median) 54 pm (aluminum cation) (lowest) 138 pm (potassium cation) (highest) cobalt(II) ions | | 65 pm (median) 65 pm (cobalt(II) cation) (lowest) 65 pm (cobalt(II) cation) (highest) d block ions | | 72 pm (median) 55 pm (iron(III) cation) (lowest) 115 pm (silver(I) cation) (highest) group 9 ions | | 61.5 pm (median) 55 pm (rhodium(V) cation) (lowest) 82 pm (iridium(III) cation) (highest) transition metal ions | 46 pm (median) 46 pm (vanadium(V) cation) (lowest) 46 pm (vanadium(V) cation) (highest) | 69 pm (median) 53 pm (manganese(IV) cation) (lowest) 115 pm (silver(I) cation) (highest) | 8-coordinate | 9-coordinate | 10-coordinate biomolecule ions | 113 pm (median) 78 pm (iron(III) cation) (lowest) 174 pm (cesium cation) (highest) | 124 pm (median) 124 pm (sodium cation) (lowest) 124 pm (sodium cation) (highest) | 153 pm (median) 123 pm (calcium cation) (lowest) 181 pm (cesium cation) (highest) cations | 112 pm (median) 78 pm (iron(III) cation) (lowest) 151 pm (potassium cation) (highest) | 124 pm (median) 124 pm (sodium cation) (lowest) 124 pm (sodium cation) (highest) | 131.5 pm (median) 123 pm (calcium cation) (lowest) 140 pm (lead(II) cation) (highest) cobalt(II) ions | 90 pm (median) 90 pm (cobalt(II) cation) (lowest) 90 pm (cobalt(II) cation) (highest) | | d block ions | 87 pm (median) 74 pm (titanium(IV) cation) (lowest) 128 pm (silver(I) cation) (highest) | 89 pm (median) 89 pm (zirconium(IV) cation) (lowest) 89 pm (zirconium(IV) cation) (highest) | group 9 ions | 90 pm (median) 90 pm (cobalt(II) cation) (lowest) 90 pm (cobalt(II) cation) (highest) | | transition metal ions | 91 pm (median) 74 pm (titanium(IV) cation) (lowest) 128 pm (silver(I) cation) (highest) | | | 12-coordinate | thermochemical radius biomolecule ions | 161 pm (median) 131 pm (cadmium cation) (lowest) 188 pm (cesium cation) (highest) | 191 pm (median) 133 pm (hydroxide anion) (lowest) 258 pm (sulfate anion) (highest) cations | 144 pm (median) 134 pm (calcium cation) (lowest) 164 pm (potassium cation) (highest) | 137 pm (median) 137 pm (ammonium cation) (lowest) 137 pm (ammonium cation) (highest) cobalt(II) ions | | 248 pm (median) 234 pm (hexaaquacobalt(II) cation) (lowest) 319 pm (tetrachlorocobaltate(II) anion) (highest) d block ions | 131 pm (median) 131 pm (cadmium cation) (lowest) 131 pm (cadmium cation) (highest) | transition metal ions | 131 pm (median) 131 pm (cadmium cation) (lowest) 131 pm (cadmium cation) (highest) |