Input interpretation

nitric acid + aluminum ⟶ water + nitrogen + aluminum nitrate
Balanced equation

Balance the chemical equation algebraically: + ⟶ + + Add stoichiometric coefficients, c_i, to the reactants and products: c_1 + c_2 ⟶ c_3 + c_4 + c_5 Set the number of atoms in the reactants equal to the number of atoms in the products for H, N, O and Al: H: | c_1 = 2 c_3 N: | c_1 = 2 c_4 + 3 c_5 O: | 3 c_1 = c_3 + 9 c_5 Al: | c_2 = c_5 Since the coefficients are relative quantities and underdetermined, choose a coefficient to set arbitrarily. To keep the coefficients small, the arbitrary value is ordinarily one. For instance, set c_4 = 1 and solve the system of equations for the remaining coefficients: c_1 = 12 c_2 = 10/3 c_3 = 6 c_4 = 1 c_5 = 10/3 Multiply by the least common denominator, 3, to eliminate fractional coefficients: c_1 = 36 c_2 = 10 c_3 = 18 c_4 = 3 c_5 = 10 Substitute the coefficients into the chemical reaction to obtain the balanced equation: Answer: | | 36 + 10 ⟶ 18 + 3 + 10
Structures
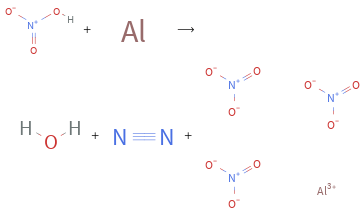
+ ⟶ + +
Names
nitric acid + aluminum ⟶ water + nitrogen + aluminum nitrate
Chemical names and formulas
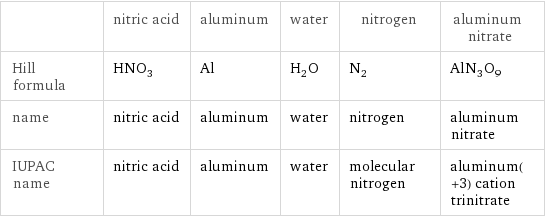
| nitric acid | aluminum | water | nitrogen | aluminum nitrate Hill formula | HNO_3 | Al | H_2O | N_2 | AlN_3O_9 name | nitric acid | aluminum | water | nitrogen | aluminum nitrate IUPAC name | nitric acid | aluminum | water | molecular nitrogen | aluminum(+3) cation trinitrate