Input interpretation
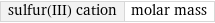
sulfur(III) cation | molar mass
Result
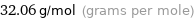
32.06 g/mol (grams per mole)
Unit conversion
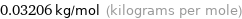
0.03206 kg/mol (kilograms per mole)
Comparisons
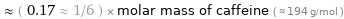
≈ ( 0.17 ≈ 1/6 ) × molar mass of caffeine ( ≈ 194 g/mol )
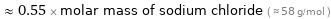
≈ 0.55 × molar mass of sodium chloride ( ≈ 58 g/mol )
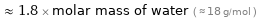
≈ 1.8 × molar mass of water ( ≈ 18 g/mol )
Corresponding quantities
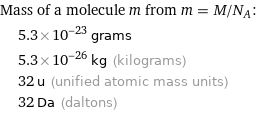
Mass of a molecule m from m = M/N_A: | 5.3×10^-23 grams | 5.3×10^-26 kg (kilograms) | 32 u (unified atomic mass units) | 32 Da (daltons)
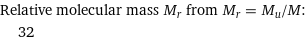
Relative molecular mass M_r from M_r = M_u/M: | 32