Input interpretation
rubidium bromide | rubidium fluoride
Chemical names and formulas

| rubidium bromide | rubidium fluoride formula | RbBr | RbF Hill formula | BrRb | FRb name | rubidium bromide | rubidium fluoride IUPAC name | rubidium(+1) cation bromide | rubidium(+1) cation fluoride alternate names | hydrobromic acid rubidium salt | rubidium(+1) cation bromide | rubidium-bromid | rubidium(+1) cation fluoride | rubidium monofluoride mass fractions | Br (bromine) 48.3% | Rb (rubidium) 51.7% | F (fluorine) 18.2% | Rb (rubidium) 81.8%
Structure diagrams

Structure diagrams
Basic properties
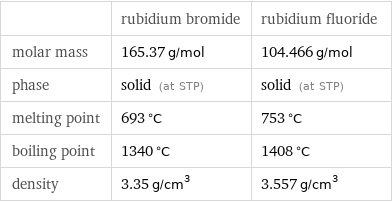
| rubidium bromide | rubidium fluoride molar mass | 165.37 g/mol | 104.466 g/mol phase | solid (at STP) | solid (at STP) melting point | 693 °C | 753 °C boiling point | 1340 °C | 1408 °C density | 3.35 g/cm^3 | 3.557 g/cm^3
Units

Thermodynamic properties
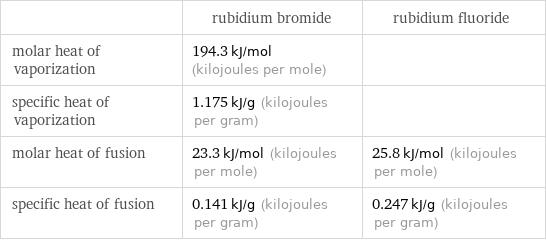
| rubidium bromide | rubidium fluoride molar heat of vaporization | 194.3 kJ/mol (kilojoules per mole) | specific heat of vaporization | 1.175 kJ/g (kilojoules per gram) | molar heat of fusion | 23.3 kJ/mol (kilojoules per mole) | 25.8 kJ/mol (kilojoules per mole) specific heat of fusion | 0.141 kJ/g (kilojoules per gram) | 0.247 kJ/g (kilojoules per gram)