Input interpretation

group 2 elements | atomic radius
Summary
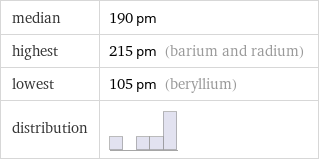
median | 190 pm highest | 215 pm (barium and radium) lowest | 105 pm (beryllium) distribution |
Units

Ranked values
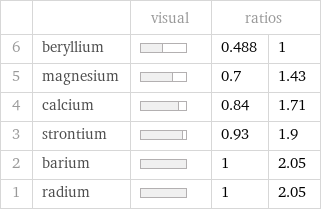
| | visual | ratios | 6 | beryllium | | 0.488 | 1 5 | magnesium | | 0.7 | 1.43 4 | calcium | | 0.84 | 1.71 3 | strontium | | 0.93 | 1.9 2 | barium | | 1 | 2.05 1 | radium | | 1 | 2.05
Plot
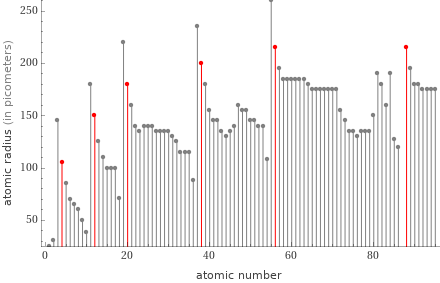
Plot
Periodic table location
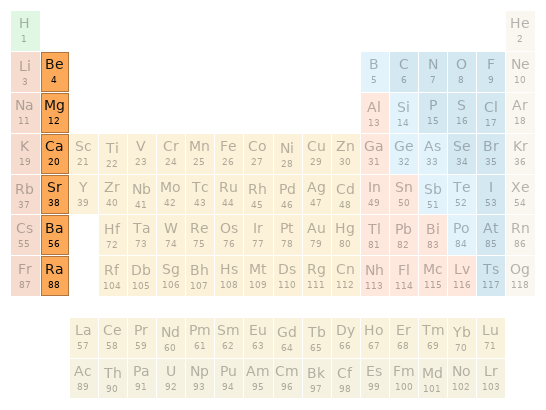
Periodic table location
Atomic radii properties

atomic radius (empirical) | median | 190 pm | highest | 215 pm (barium and radium) | lowest | 105 pm (beryllium) | distribution | atomic radius (calculated) | median | 194 pm | highest | 253 pm (barium) | lowest | 112 pm (beryllium) | distribution | covalent radius | median | 186 pm | highest | 221 pm (radium) | lowest | 96 pm (beryllium) | distribution |
Units

Atomic radius rankings
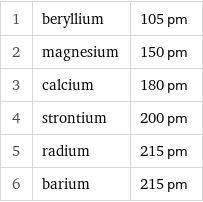
1 | beryllium | 105 pm 2 | magnesium | 150 pm 3 | calcium | 180 pm 4 | strontium | 200 pm 5 | radium | 215 pm 6 | barium | 215 pm
Unit conversions for median atomic radius 190 pm
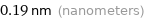
0.19 nm (nanometers)
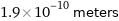
1.9×10^-10 meters
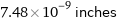
7.48×10^-9 inches
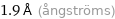
1.9 Å (ångströms)
Comparisons for median atomic radius 190 pm
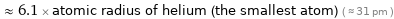
≈ 6.1 × atomic radius of helium (the smallest atom) ( ≈ 31 pm )
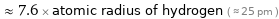
≈ 7.6 × atomic radius of hydrogen ( ≈ 25 pm )