Input interpretation
fluoromethane
Chemical names and formulas
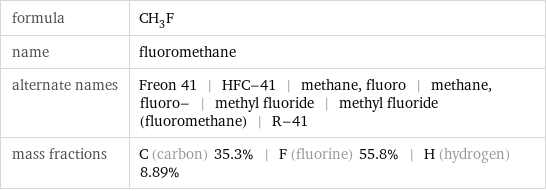
formula | CH_3F name | fluoromethane alternate names | Freon 41 | HFC-41 | methane, fluoro | methane, fluoro- | methyl fluoride | methyl fluoride (fluoromethane) | R-41 mass fractions | C (carbon) 35.3% | F (fluorine) 55.8% | H (hydrogen) 8.89%
Lewis structure

Draw the Lewis structure of fluoromethane. Start by drawing the overall structure of the molecule: Count the total valence electrons of the carbon (n_C, val = 4), fluorine (n_F, val = 7), and hydrogen (n_H, val = 1) atoms: n_C, val + n_F, val + 3 n_H, val = 14 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), fluorine (n_F, full = 8), and hydrogen (n_H, full = 2): n_C, full + n_F, full + 3 n_H, full = 22 Subtracting these two numbers shows that 22 - 14 = 8 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 4 bonds and hence 8 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 14 - 8 = 6 electrons left to draw: Answer: | |
3D structure

3D structure
Basic properties
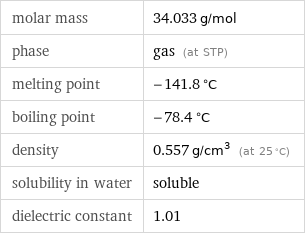
molar mass | 34.033 g/mol phase | gas (at STP) melting point | -141.8 °C boiling point | -78.4 °C density | 0.557 g/cm^3 (at 25 °C) solubility in water | soluble dielectric constant | 1.01
Gas properties (at STP)
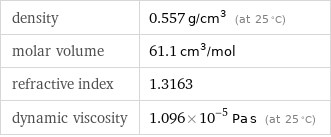
density | 0.557 g/cm^3 (at 25 °C) molar volume | 61.1 cm^3/mol refractive index | 1.3163 dynamic viscosity | 1.096×10^-5 Pa s (at 25 °C)
Units
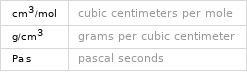
Thermodynamic properties

specific heat capacity c_p | gas | 1.102 J/(g K) molar heat capacity c_p | gas | 37.5 J/(mol K) specific entropy S° | gas | 6.549 J/(g K) molar entropy S° | gas | 222.9 J/(mol K) molar heat of vaporization | 17.2 kJ/mol | specific heat of vaporization | 0.505 kJ/g | critical temperature | 320 K | critical pressure | 5.877 MPa | (at STP)
Phase diagram
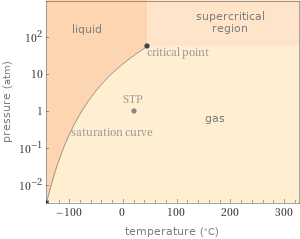
Phase diagram
Units

Chemical identifiers
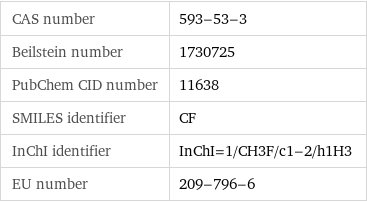
CAS number | 593-53-3 Beilstein number | 1730725 PubChem CID number | 11638 SMILES identifier | CF InChI identifier | InChI=1/CH3F/c1-2/h1H3 EU number | 209-796-6
Safety properties
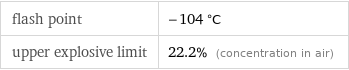
flash point | -104 °C upper explosive limit | 22.2% (concentration in air)

DOT hazard class | 2.1 DOT numbers | 2454