Input interpretation
pyrite | chromium(III) hydroxide trihydrate
Chemical names and formulas
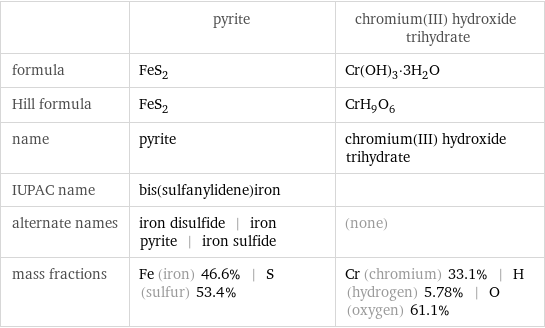
| pyrite | chromium(III) hydroxide trihydrate formula | FeS_2 | Cr(OH)_3·3H_2O Hill formula | FeS_2 | CrH_9O_6 name | pyrite | chromium(III) hydroxide trihydrate IUPAC name | bis(sulfanylidene)iron | alternate names | iron disulfide | iron pyrite | iron sulfide | (none) mass fractions | Fe (iron) 46.6% | S (sulfur) 53.4% | Cr (chromium) 33.1% | H (hydrogen) 5.78% | O (oxygen) 61.1%
Structure diagrams
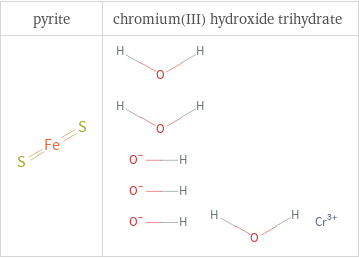
Structure diagrams
Basic properties
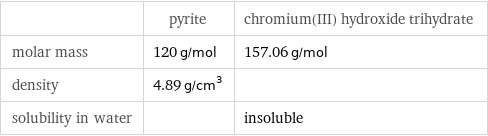
| pyrite | chromium(III) hydroxide trihydrate molar mass | 120 g/mol | 157.06 g/mol density | 4.89 g/cm^3 | solubility in water | | insoluble
Units
