Input interpretation
zinc chloride | calcium chloride dihydrate
Chemical names and formulas
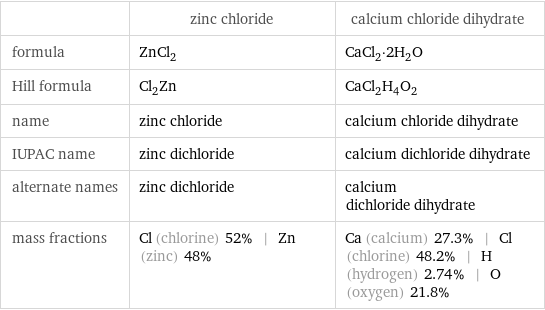
| zinc chloride | calcium chloride dihydrate formula | ZnCl_2 | CaCl_2·2H_2O Hill formula | Cl_2Zn | CaCl_2H_4O_2 name | zinc chloride | calcium chloride dihydrate IUPAC name | zinc dichloride | calcium dichloride dihydrate alternate names | zinc dichloride | calcium dichloride dihydrate mass fractions | Cl (chlorine) 52% | Zn (zinc) 48% | Ca (calcium) 27.3% | Cl (chlorine) 48.2% | H (hydrogen) 2.74% | O (oxygen) 21.8%
Structure diagrams

Structure diagrams
Basic properties
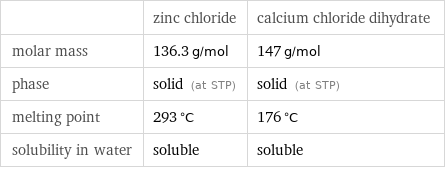
| zinc chloride | calcium chloride dihydrate molar mass | 136.3 g/mol | 147 g/mol phase | solid (at STP) | solid (at STP) melting point | 293 °C | 176 °C solubility in water | soluble | soluble
Units
