Input interpretation

greenhouse gases | molar heat of combustion
Summary

median | 1166 kJ/mol highest | 6551 kJ/mol (methyl chloride) lowest | 156.2 kJ/mol (carbon tetrachloride) | (based on 4 values; 17 unavailable)
Entities with missing values
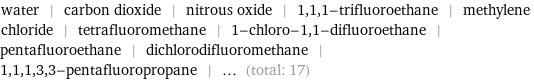
water | carbon dioxide | nitrous oxide | 1, 1, 1-trifluoroethane | methylene chloride | tetrafluoromethane | 1-chloro-1, 1-difluoroethane | pentafluoroethane | dichlorodifluoromethane | 1, 1, 1, 3, 3-pentafluoropropane | ... (total: 17)
Molar heat of combustion rankings
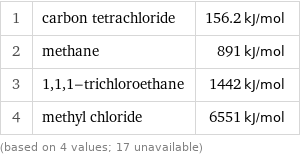
1 | carbon tetrachloride | 156.2 kJ/mol 2 | methane | 891 kJ/mol 3 | 1, 1, 1-trichloroethane | 1442 kJ/mol 4 | methyl chloride | 6551 kJ/mol (based on 4 values; 17 unavailable)
Unit conversions for median molar heat of combustion 1166 kJ/mol
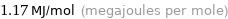
1.17 MJ/mol (megajoules per mole)
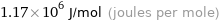
1.17×10^6 J/mol (joules per mole)
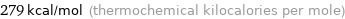
279 kcal/mol (thermochemical kilocalories per mole)
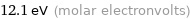
12.1 eV (molar electronvolts)
Corresponding quantity

Spectroscopic wavenumber ν^~: | 97513 cm^(-1) (reciprocal centimeters)