Input interpretation
ceric oxide
Chemical names and formulas

formula | CeO_2 name | ceric oxide IUPAC name | dioxocerium alternate names | ceria | cerium dioxide | cerium(IV) oxide | cerium oxide | diketocerium | dioxocerium mass fractions | Ce (cerium) 81.4% | O (oxygen) 18.6%
Structure diagram
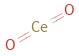
Structure diagram
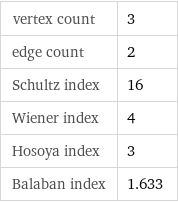
vertex count | 3 edge count | 2 Schultz index | 16 Wiener index | 4 Hosoya index | 3 Balaban index | 1.633
Basic properties
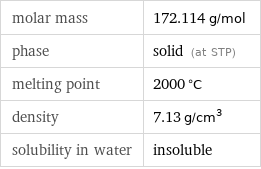
molar mass | 172.114 g/mol phase | solid (at STP) melting point | 2000 °C density | 7.13 g/cm^3 solubility in water | insoluble
Units

Solid properties (at STP)

density | 7.13 g/cm^3
Units

Thermodynamic properties
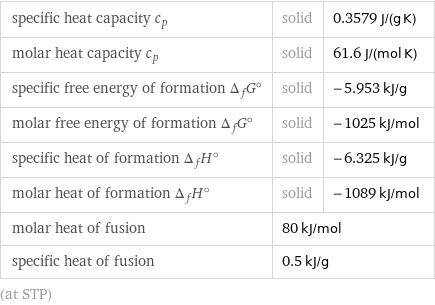
specific heat capacity c_p | solid | 0.3579 J/(g K) molar heat capacity c_p | solid | 61.6 J/(mol K) specific free energy of formation Δ_fG° | solid | -5.953 kJ/g molar free energy of formation Δ_fG° | solid | -1025 kJ/mol specific heat of formation Δ_fH° | solid | -6.325 kJ/g molar heat of formation Δ_fH° | solid | -1089 kJ/mol molar heat of fusion | 80 kJ/mol | specific heat of fusion | 0.5 kJ/g | (at STP)
Chemical identifiers
![CAS number | 1306-38-3 PubChem CID number | 73963 PubChem SID number | 24852135 SMILES identifier | O=[Ce]=O InChI identifier | InChI=1/Ce.2O/rCeO2/c2-1-3 RTECS number | FK6310000 MDL number | MFCD00010933](../image_source/48cbff58ace1172d5c0ea6b6a228d6ee.png)
CAS number | 1306-38-3 PubChem CID number | 73963 PubChem SID number | 24852135 SMILES identifier | O=[Ce]=O InChI identifier | InChI=1/Ce.2O/rCeO2/c2-1-3 RTECS number | FK6310000 MDL number | MFCD00010933
NFPA label

NFPA label

NFPA health rating | 1 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties

RTECS classes | other