Input interpretation
group 13 elements | number of valence electrons
Summary
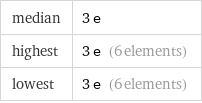
median | 3 e highest | 3 e (6 elements) lowest | 3 e (6 elements)
Units

Ranked values
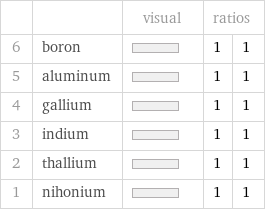
| | visual | ratios | 6 | boron | | 1 | 1 5 | aluminum | | 1 | 1 4 | gallium | | 1 | 1 3 | indium | | 1 | 1 2 | thallium | | 1 | 1 1 | nihonium | | 1 | 1
Number of valence electrons rankings
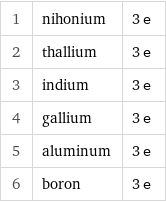
1 | nihonium | 3 e 2 | thallium | 3 e 3 | indium | 3 e 4 | gallium | 3 e 5 | aluminum | 3 e 6 | boron | 3 e
Corresponding quantity
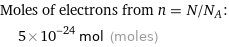
Moles of electrons from n = N/N_A: | 5×10^-24 mol (moles)