Input interpretation
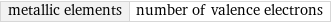
metallic elements | number of valence electrons
Summary

median | 2 e highest | 10 e (palladium) lowest | 1 e (17 elements) distribution |
Units

Number of valence electrons rankings

1 | roentgenium | 1 e 2 | darmstadtium | 1 e 3 | francium | 1 e 4 | gold | 1 e 5 | platinum | 1 e 6 | cesium | 1 e 7 | silver | 1 e 8 | rhodium | 1 e 9 | ruthenium | 1 e 10 | molybdenum | 1 e ⋮ | | 85 | flerovium | 4 e 86 | lead | 4 e 87 | tin | 4 e 88 | moscovium | 5 e 89 | bismuth | 5 e 90 | antimony | 5 e 91 | arsenic | 5 e 92 | livermorium | 6 e 93 | polonium | 6 e 94 | palladium | 10 e
Corresponding quantity
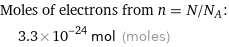
Moles of electrons from n = N/N_A: | 3.3×10^-24 mol (moles)