Input interpretation
zirconium (chemical element) | iodine (chemical element) | manganese (chemical element)
Periodic table location
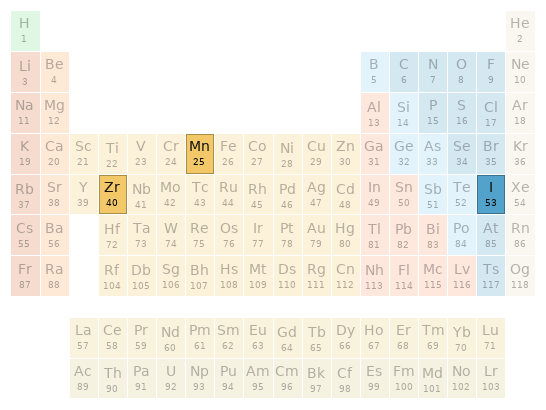
Periodic table location
Images

Images
Basic elemental properties
![| zirconium | iodine | manganese atomic symbol | Zr | I | Mn atomic number | 40 | 53 | 25 short electronic configuration | [Kr]5s^24d^2 | [Kr]5s^24d^105p^5 | [Ar]4s^23d^5 Aufbau diagram | 4d 5s | 5p 4d 5s | 3d 4s block | d | p | d group | 4 | 17 | 7 period | 5 | 5 | 4 atomic mass | 91.224 u | 126.90447 u | 54.938044 u half-life | (stable) | (stable) | (stable)](../image_source/041ed74709af13e4291095753e6694fa.png)
| zirconium | iodine | manganese atomic symbol | Zr | I | Mn atomic number | 40 | 53 | 25 short electronic configuration | [Kr]5s^24d^2 | [Kr]5s^24d^105p^5 | [Ar]4s^23d^5 Aufbau diagram | 4d 5s | 5p 4d 5s | 3d 4s block | d | p | d group | 4 | 17 | 7 period | 5 | 5 | 4 atomic mass | 91.224 u | 126.90447 u | 54.938044 u half-life | (stable) | (stable) | (stable)
Thermodynamic properties
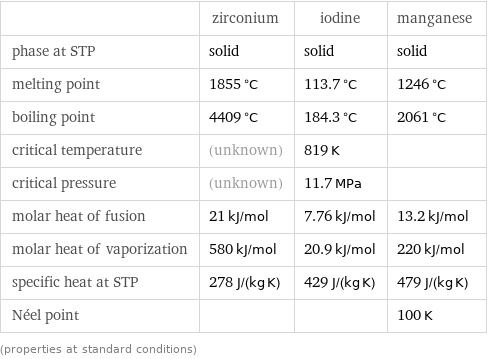
| zirconium | iodine | manganese phase at STP | solid | solid | solid melting point | 1855 °C | 113.7 °C | 1246 °C boiling point | 4409 °C | 184.3 °C | 2061 °C critical temperature | (unknown) | 819 K | critical pressure | (unknown) | 11.7 MPa | molar heat of fusion | 21 kJ/mol | 7.76 kJ/mol | 13.2 kJ/mol molar heat of vaporization | 580 kJ/mol | 20.9 kJ/mol | 220 kJ/mol specific heat at STP | 278 J/(kg K) | 429 J/(kg K) | 479 J/(kg K) Néel point | | | 100 K (properties at standard conditions)
Material properties
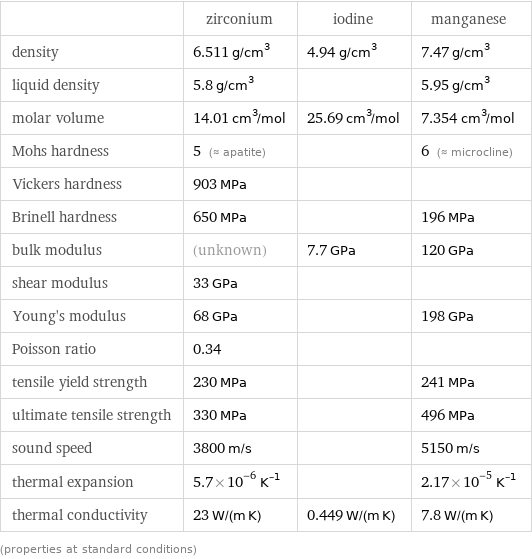
| zirconium | iodine | manganese density | 6.511 g/cm^3 | 4.94 g/cm^3 | 7.47 g/cm^3 liquid density | 5.8 g/cm^3 | | 5.95 g/cm^3 molar volume | 14.01 cm^3/mol | 25.69 cm^3/mol | 7.354 cm^3/mol Mohs hardness | 5 (≈ apatite) | | 6 (≈ microcline) Vickers hardness | 903 MPa | | Brinell hardness | 650 MPa | | 196 MPa bulk modulus | (unknown) | 7.7 GPa | 120 GPa shear modulus | 33 GPa | | Young's modulus | 68 GPa | | 198 GPa Poisson ratio | 0.34 | | tensile yield strength | 230 MPa | | 241 MPa ultimate tensile strength | 330 MPa | | 496 MPa sound speed | 3800 m/s | | 5150 m/s thermal expansion | 5.7×10^-6 K^(-1) | | 2.17×10^-5 K^(-1) thermal conductivity | 23 W/(m K) | 0.449 W/(m K) | 7.8 W/(m K) (properties at standard conditions)
Electromagnetic properties
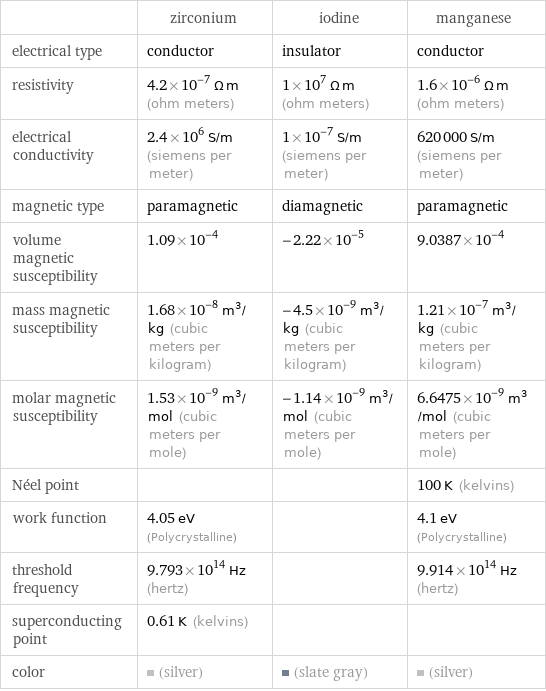
| zirconium | iodine | manganese electrical type | conductor | insulator | conductor resistivity | 4.2×10^-7 Ω m (ohm meters) | 1×10^7 Ω m (ohm meters) | 1.6×10^-6 Ω m (ohm meters) electrical conductivity | 2.4×10^6 S/m (siemens per meter) | 1×10^-7 S/m (siemens per meter) | 620000 S/m (siemens per meter) magnetic type | paramagnetic | diamagnetic | paramagnetic volume magnetic susceptibility | 1.09×10^-4 | -2.22×10^-5 | 9.0387×10^-4 mass magnetic susceptibility | 1.68×10^-8 m^3/kg (cubic meters per kilogram) | -4.5×10^-9 m^3/kg (cubic meters per kilogram) | 1.21×10^-7 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | 1.53×10^-9 m^3/mol (cubic meters per mole) | -1.14×10^-9 m^3/mol (cubic meters per mole) | 6.6475×10^-9 m^3/mol (cubic meters per mole) Néel point | | | 100 K (kelvins) work function | 4.05 eV (Polycrystalline) | | 4.1 eV (Polycrystalline) threshold frequency | 9.793×10^14 Hz (hertz) | | 9.914×10^14 Hz (hertz) superconducting point | 0.61 K (kelvins) | | color | (silver) | (slate gray) | (silver)
Reactivity
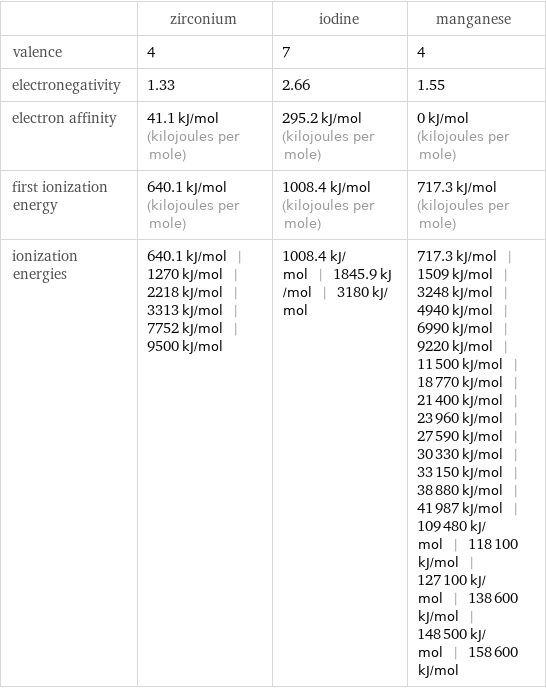
| zirconium | iodine | manganese valence | 4 | 7 | 4 electronegativity | 1.33 | 2.66 | 1.55 electron affinity | 41.1 kJ/mol (kilojoules per mole) | 295.2 kJ/mol (kilojoules per mole) | 0 kJ/mol (kilojoules per mole) first ionization energy | 640.1 kJ/mol (kilojoules per mole) | 1008.4 kJ/mol (kilojoules per mole) | 717.3 kJ/mol (kilojoules per mole) ionization energies | 640.1 kJ/mol | 1270 kJ/mol | 2218 kJ/mol | 3313 kJ/mol | 7752 kJ/mol | 9500 kJ/mol | 1008.4 kJ/mol | 1845.9 kJ/mol | 3180 kJ/mol | 717.3 kJ/mol | 1509 kJ/mol | 3248 kJ/mol | 4940 kJ/mol | 6990 kJ/mol | 9220 kJ/mol | 11500 kJ/mol | 18770 kJ/mol | 21400 kJ/mol | 23960 kJ/mol | 27590 kJ/mol | 30330 kJ/mol | 33150 kJ/mol | 38880 kJ/mol | 41987 kJ/mol | 109480 kJ/mol | 118100 kJ/mol | 127100 kJ/mol | 138600 kJ/mol | 148500 kJ/mol | 158600 kJ/mol
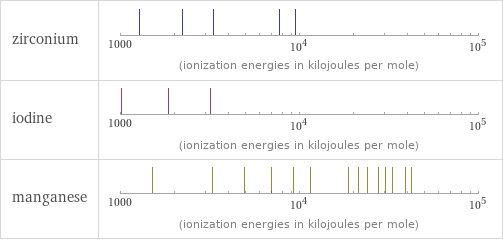
Reactivity
Atomic properties
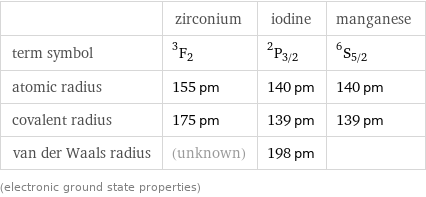
| zirconium | iodine | manganese term symbol | ^3F_2 | ^2P_(3/2) | ^6S_(5/2) atomic radius | 155 pm | 140 pm | 140 pm covalent radius | 175 pm | 139 pm | 139 pm van der Waals radius | (unknown) | 198 pm | (electronic ground state properties)
Abundances
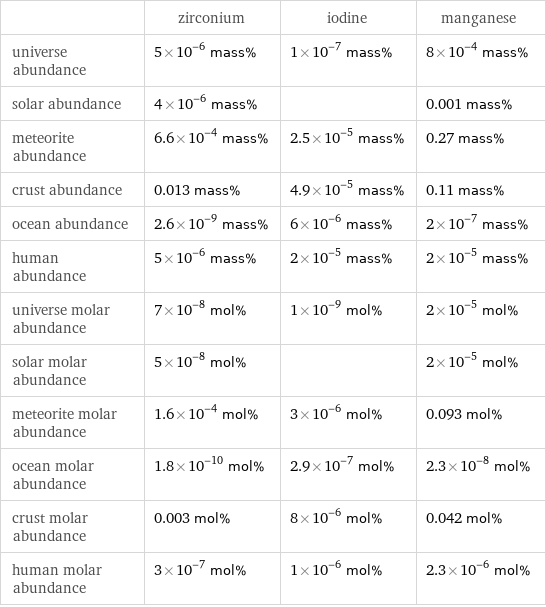
| zirconium | iodine | manganese universe abundance | 5×10^-6 mass% | 1×10^-7 mass% | 8×10^-4 mass% solar abundance | 4×10^-6 mass% | | 0.001 mass% meteorite abundance | 6.6×10^-4 mass% | 2.5×10^-5 mass% | 0.27 mass% crust abundance | 0.013 mass% | 4.9×10^-5 mass% | 0.11 mass% ocean abundance | 2.6×10^-9 mass% | 6×10^-6 mass% | 2×10^-7 mass% human abundance | 5×10^-6 mass% | 2×10^-5 mass% | 2×10^-5 mass% universe molar abundance | 7×10^-8 mol% | 1×10^-9 mol% | 2×10^-5 mol% solar molar abundance | 5×10^-8 mol% | | 2×10^-5 mol% meteorite molar abundance | 1.6×10^-4 mol% | 3×10^-6 mol% | 0.093 mol% ocean molar abundance | 1.8×10^-10 mol% | 2.9×10^-7 mol% | 2.3×10^-8 mol% crust molar abundance | 0.003 mol% | 8×10^-6 mol% | 0.042 mol% human molar abundance | 3×10^-7 mol% | 1×10^-6 mol% | 2.3×10^-6 mol%