Input interpretation

cations | molar mass
Summary
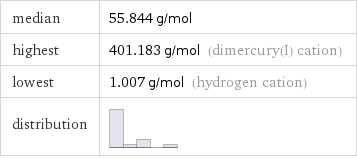
median | 55.844 g/mol highest | 401.183 g/mol (dimercury(I) cation) lowest | 1.007 g/mol (hydrogen cation) distribution |
Units

Distribution plots
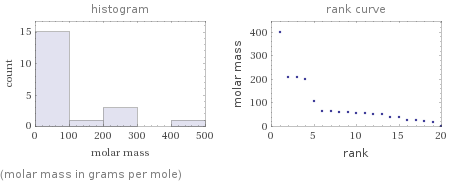
(molar mass in grams per mole)
Molar mass rankings
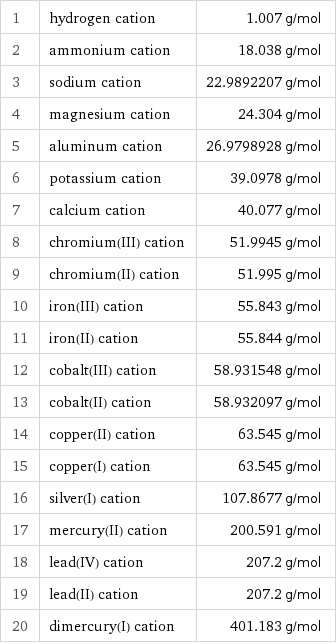
1 | hydrogen cation | 1.007 g/mol 2 | ammonium cation | 18.038 g/mol 3 | sodium cation | 22.9892207 g/mol 4 | magnesium cation | 24.304 g/mol 5 | aluminum cation | 26.9798928 g/mol 6 | potassium cation | 39.0978 g/mol 7 | calcium cation | 40.077 g/mol 8 | chromium(III) cation | 51.9945 g/mol 9 | chromium(II) cation | 51.995 g/mol 10 | iron(III) cation | 55.843 g/mol 11 | iron(II) cation | 55.844 g/mol 12 | cobalt(III) cation | 58.931548 g/mol 13 | cobalt(II) cation | 58.932097 g/mol 14 | copper(II) cation | 63.545 g/mol 15 | copper(I) cation | 63.545 g/mol 16 | silver(I) cation | 107.8677 g/mol 17 | mercury(II) cation | 200.591 g/mol 18 | lead(IV) cation | 207.2 g/mol 19 | lead(II) cation | 207.2 g/mol 20 | dimercury(I) cation | 401.183 g/mol
Unit conversion for median molar mass 55.844 g/mol
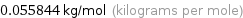
0.055844 kg/mol (kilograms per mole)
Comparisons for median molar mass 55.844 g/mol
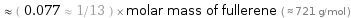
≈ ( 0.077 ≈ 1/13 ) × molar mass of fullerene ( ≈ 721 g/mol )
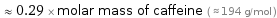
≈ 0.29 × molar mass of caffeine ( ≈ 194 g/mol )
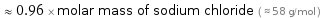
≈ 0.96 × molar mass of sodium chloride ( ≈ 58 g/mol )
Corresponding quantities
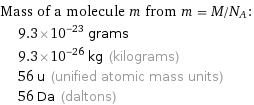
Mass of a molecule m from m = M/N_A: | 9.3×10^-23 grams | 9.3×10^-26 kg (kilograms) | 56 u (unified atomic mass units) | 56 Da (daltons)
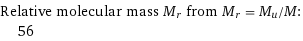
Relative molecular mass M_r from M_r = M_u/M: | 56