Input interpretation
group 18 elements
Members
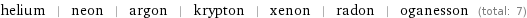
helium | neon | argon | krypton | xenon | radon | oganesson (total: 7)
Periodic table location
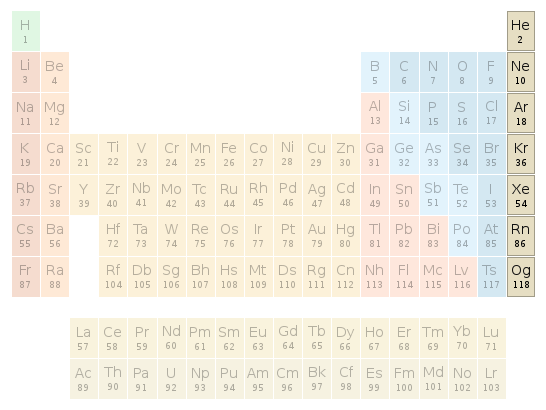
Periodic table location
Images

Images
Basic elemental properties

atomic symbol | all | Ar | He | Kr | Ne | Og | Rn | Xe atomic number | median | 36 | highest | 118 (oganesson) | lowest | 2 (helium) | distribution | block | all | p | s group | all | 18 period | all | 1 | 2 | 3 | 4 | 5 | 6 | 7 atomic mass | median | 83.798 u | highest | 294 u (oganesson) | lowest | 4.002602 u (helium) | distribution |
Thermodynamic properties
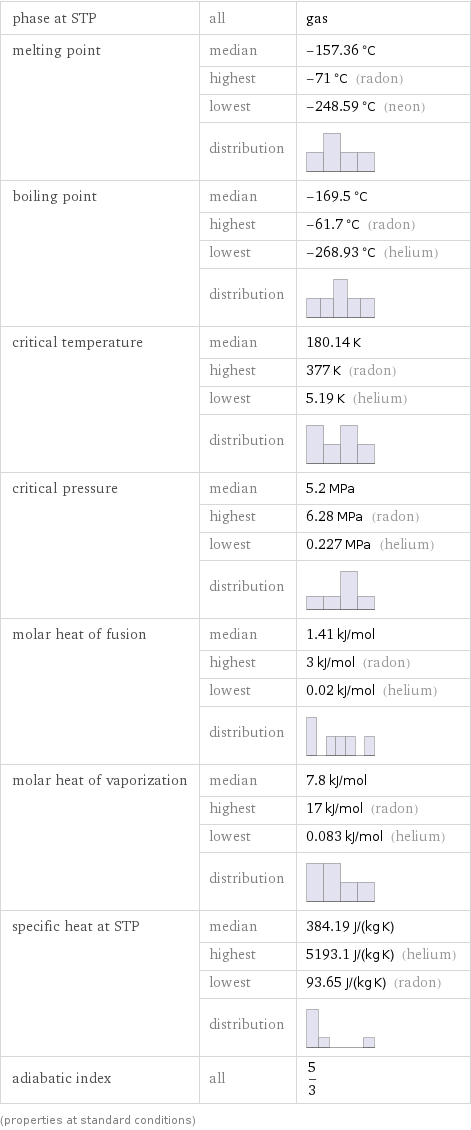
phase at STP | all | gas melting point | median | -157.36 °C | highest | -71 °C (radon) | lowest | -248.59 °C (neon) | distribution | boiling point | median | -169.5 °C | highest | -61.7 °C (radon) | lowest | -268.93 °C (helium) | distribution | critical temperature | median | 180.14 K | highest | 377 K (radon) | lowest | 5.19 K (helium) | distribution | critical pressure | median | 5.2 MPa | highest | 6.28 MPa (radon) | lowest | 0.227 MPa (helium) | distribution | molar heat of fusion | median | 1.41 kJ/mol | highest | 3 kJ/mol (radon) | lowest | 0.02 kJ/mol (helium) | distribution | molar heat of vaporization | median | 7.8 kJ/mol | highest | 17 kJ/mol (radon) | lowest | 0.083 kJ/mol (helium) | distribution | specific heat at STP | median | 384.19 J/(kg K) | highest | 5193.1 J/(kg K) (helium) | lowest | 93.65 J/(kg K) (radon) | distribution | adiabatic index | all | 5/3 (properties at standard conditions)
Material properties
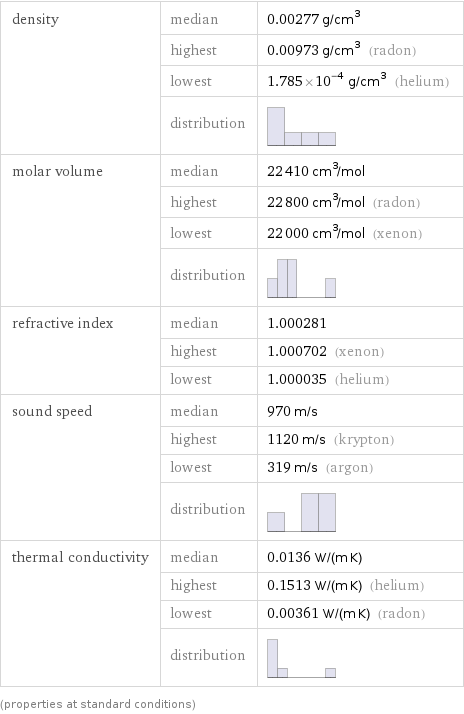
density | median | 0.00277 g/cm^3 | highest | 0.00973 g/cm^3 (radon) | lowest | 1.785×10^-4 g/cm^3 (helium) | distribution | molar volume | median | 22410 cm^3/mol | highest | 22800 cm^3/mol (radon) | lowest | 22000 cm^3/mol (xenon) | distribution | refractive index | median | 1.000281 | highest | 1.000702 (xenon) | lowest | 1.000035 (helium) sound speed | median | 970 m/s | highest | 1120 m/s (krypton) | lowest | 319 m/s (argon) | distribution | thermal conductivity | median | 0.0136 W/(m K) | highest | 0.1513 W/(m K) (helium) | lowest | 0.00361 W/(m K) (radon) | distribution | (properties at standard conditions)
Reactivity
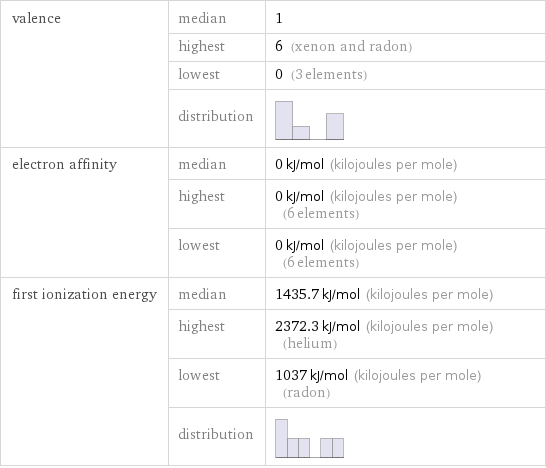
valence | median | 1 | highest | 6 (xenon and radon) | lowest | 0 (3 elements) | distribution | electron affinity | median | 0 kJ/mol (kilojoules per mole) | highest | 0 kJ/mol (kilojoules per mole) (6 elements) | lowest | 0 kJ/mol (kilojoules per mole) (6 elements) first ionization energy | median | 1435.7 kJ/mol (kilojoules per mole) | highest | 2372.3 kJ/mol (kilojoules per mole) (helium) | lowest | 1037 kJ/mol (kilojoules per mole) (radon) | distribution |
Atomic properties
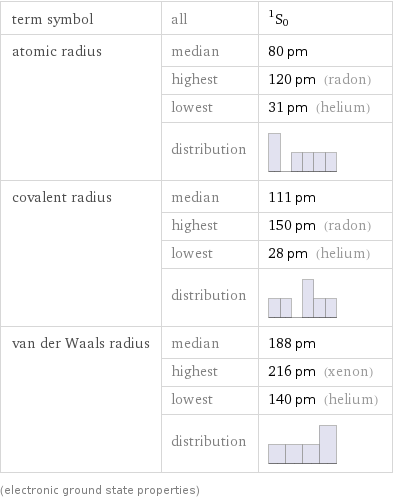
term symbol | all | ^1S_0 atomic radius | median | 80 pm | highest | 120 pm (radon) | lowest | 31 pm (helium) | distribution | covalent radius | median | 111 pm | highest | 150 pm (radon) | lowest | 28 pm (helium) | distribution | van der Waals radius | median | 188 pm | highest | 216 pm (xenon) | lowest | 140 pm (helium) | distribution | (electronic ground state properties)
Abundances
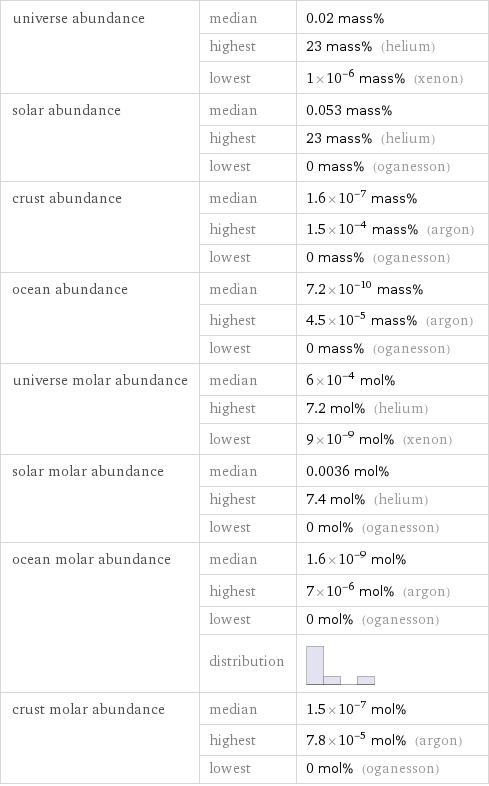
universe abundance | median | 0.02 mass% | highest | 23 mass% (helium) | lowest | 1×10^-6 mass% (xenon) solar abundance | median | 0.053 mass% | highest | 23 mass% (helium) | lowest | 0 mass% (oganesson) crust abundance | median | 1.6×10^-7 mass% | highest | 1.5×10^-4 mass% (argon) | lowest | 0 mass% (oganesson) ocean abundance | median | 7.2×10^-10 mass% | highest | 4.5×10^-5 mass% (argon) | lowest | 0 mass% (oganesson) universe molar abundance | median | 6×10^-4 mol% | highest | 7.2 mol% (helium) | lowest | 9×10^-9 mol% (xenon) solar molar abundance | median | 0.0036 mol% | highest | 7.4 mol% (helium) | lowest | 0 mol% (oganesson) ocean molar abundance | median | 1.6×10^-9 mol% | highest | 7×10^-6 mol% (argon) | lowest | 0 mol% (oganesson) | distribution | crust molar abundance | median | 1.5×10^-7 mol% | highest | 7.8×10^-5 mol% (argon) | lowest | 0 mol% (oganesson)