Input interpretation

triparanol | element types
Result
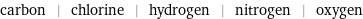
carbon | chlorine | hydrogen | nitrogen | oxygen
Periodic table location
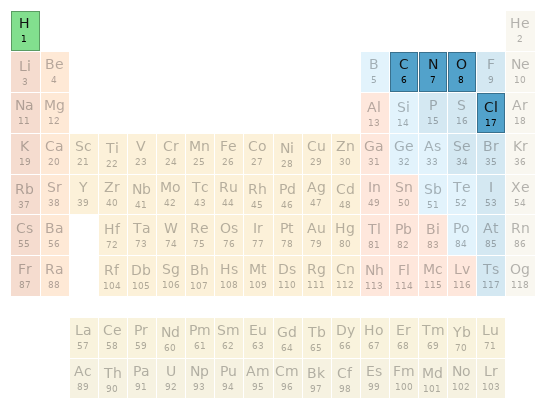
Periodic table location
Images

Images
Basic elemental properties
![| atomic symbol | atomic number | short electronic configuration carbon | C | 6 | [He]2s^22p^2 chlorine | Cl | 17 | [Ne]3s^23p^5 hydrogen | H | 1 | 1s^1 nitrogen | N | 7 | [He]2s^22p^3 oxygen | O | 8 | [He]2s^22p^4 | Aufbau diagram | block | group carbon | 2p 2s | p | 14 chlorine | 3p 3s | p | 17 hydrogen | 1s | s | 1 nitrogen | 2p 2s | p | 15 oxygen | 2p 2s | p | 16 | period | atomic mass | half-life carbon | 2 | 12.011 u | (stable) chlorine | 3 | 35.45 u | (stable) hydrogen | 1 | 1.008 u | (stable) nitrogen | 2 | 14.007 u | (stable) oxygen | 2 | 15.999 u | (stable)](../image_source/3d82171eb9cc1e032dc4147f582efad6.png)
| atomic symbol | atomic number | short electronic configuration carbon | C | 6 | [He]2s^22p^2 chlorine | Cl | 17 | [Ne]3s^23p^5 hydrogen | H | 1 | 1s^1 nitrogen | N | 7 | [He]2s^22p^3 oxygen | O | 8 | [He]2s^22p^4 | Aufbau diagram | block | group carbon | 2p 2s | p | 14 chlorine | 3p 3s | p | 17 hydrogen | 1s | s | 1 nitrogen | 2p 2s | p | 15 oxygen | 2p 2s | p | 16 | period | atomic mass | half-life carbon | 2 | 12.011 u | (stable) chlorine | 3 | 35.45 u | (stable) hydrogen | 1 | 1.008 u | (stable) nitrogen | 2 | 14.007 u | (stable) oxygen | 2 | 15.999 u | (stable)
Thermodynamic properties
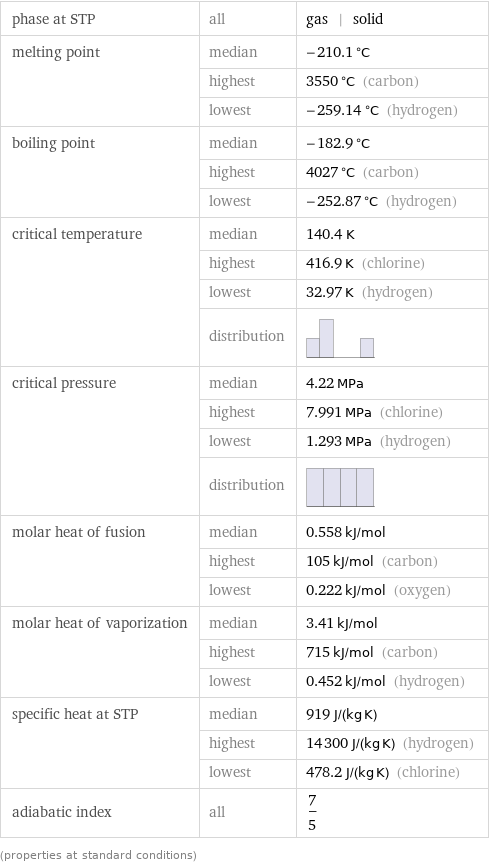
phase at STP | all | gas | solid melting point | median | -210.1 °C | highest | 3550 °C (carbon) | lowest | -259.14 °C (hydrogen) boiling point | median | -182.9 °C | highest | 4027 °C (carbon) | lowest | -252.87 °C (hydrogen) critical temperature | median | 140.4 K | highest | 416.9 K (chlorine) | lowest | 32.97 K (hydrogen) | distribution | critical pressure | median | 4.22 MPa | highest | 7.991 MPa (chlorine) | lowest | 1.293 MPa (hydrogen) | distribution | molar heat of fusion | median | 0.558 kJ/mol | highest | 105 kJ/mol (carbon) | lowest | 0.222 kJ/mol (oxygen) molar heat of vaporization | median | 3.41 kJ/mol | highest | 715 kJ/mol (carbon) | lowest | 0.452 kJ/mol (hydrogen) specific heat at STP | median | 919 J/(kg K) | highest | 14300 J/(kg K) (hydrogen) | lowest | 478.2 J/(kg K) (chlorine) adiabatic index | all | 7/5 (properties at standard conditions)
Material properties
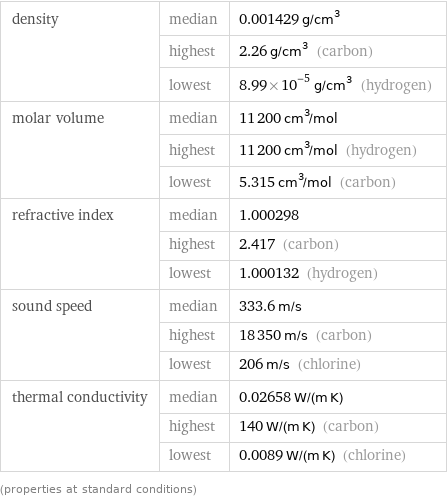
density | median | 0.001429 g/cm^3 | highest | 2.26 g/cm^3 (carbon) | lowest | 8.99×10^-5 g/cm^3 (hydrogen) molar volume | median | 11200 cm^3/mol | highest | 11200 cm^3/mol (hydrogen) | lowest | 5.315 cm^3/mol (carbon) refractive index | median | 1.000298 | highest | 2.417 (carbon) | lowest | 1.000132 (hydrogen) sound speed | median | 333.6 m/s | highest | 18350 m/s (carbon) | lowest | 206 m/s (chlorine) thermal conductivity | median | 0.02658 W/(m K) | highest | 140 W/(m K) (carbon) | lowest | 0.0089 W/(m K) (chlorine) (properties at standard conditions)
Reactivity

valence | median | 3 | highest | 5 (chlorine) | lowest | 1 (hydrogen) | distribution | electronegativity | median | 3.04 | highest | 3.44 (oxygen) | lowest | 2.2 (hydrogen) | distribution | electron affinity | median | 141 kJ/mol (kilojoules per mole) | highest | 349 kJ/mol (kilojoules per mole) (chlorine) | lowest | 7 kJ/mol (kilojoules per mole) (nitrogen) | distribution | first ionization energy | median | 1312 kJ/mol (kilojoules per mole) | highest | 1402.3 kJ/mol (kilojoules per mole) (nitrogen) | lowest | 1086.5 kJ/mol (kilojoules per mole) (carbon) | distribution |