Input interpretation
nickel(II) fluoborate | strontium hydroxide octahydrate
Chemical names and formulas
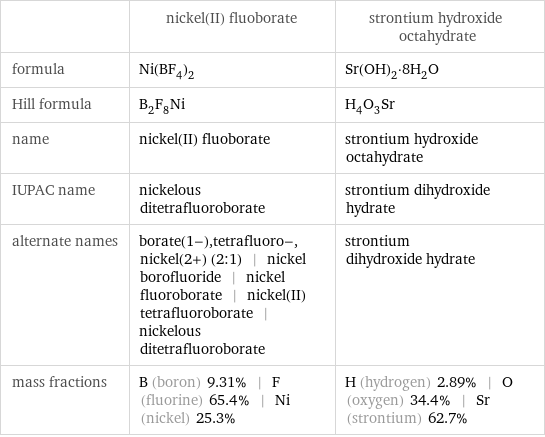
| nickel(II) fluoborate | strontium hydroxide octahydrate formula | Ni(BF_4)_2 | Sr(OH)_2·8H_2O Hill formula | B_2F_8Ni | H_4O_3Sr name | nickel(II) fluoborate | strontium hydroxide octahydrate IUPAC name | nickelous ditetrafluoroborate | strontium dihydroxide hydrate alternate names | borate(1-), tetrafluoro-, nickel(2+) (2:1) | nickel borofluoride | nickel fluoroborate | nickel(II) tetrafluoroborate | nickelous ditetrafluoroborate | strontium dihydroxide hydrate mass fractions | B (boron) 9.31% | F (fluorine) 65.4% | Ni (nickel) 25.3% | H (hydrogen) 2.89% | O (oxygen) 34.4% | Sr (strontium) 62.7%
Structure diagrams
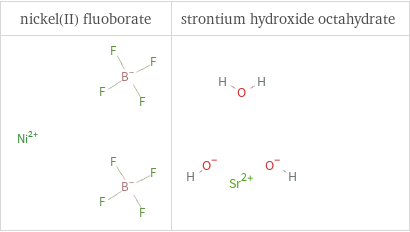
Structure diagrams
Basic properties

| nickel(II) fluoborate | strontium hydroxide octahydrate molar mass | 232.3 g/mol | 139.6 g/mol phase | liquid (at STP) | boiling point | 100 °C | density | 1.454 g/cm^3 | 1.9 g/cm^3
Units

Liquid properties
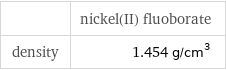
| nickel(II) fluoborate density | 1.454 g/cm^3
Units
