Input interpretation
magnesium sulfide | cerous chloride
Chemical names and formulas
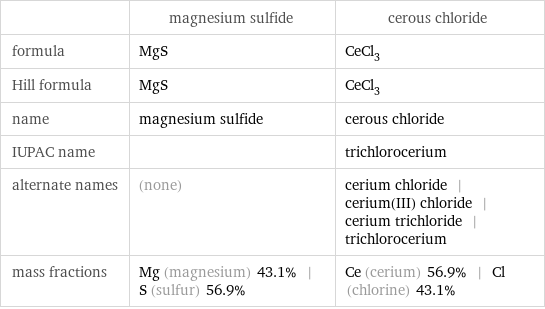
| magnesium sulfide | cerous chloride formula | MgS | CeCl_3 Hill formula | MgS | CeCl_3 name | magnesium sulfide | cerous chloride IUPAC name | | trichlorocerium alternate names | (none) | cerium chloride | cerium(III) chloride | cerium trichloride | trichlorocerium mass fractions | Mg (magnesium) 43.1% | S (sulfur) 56.9% | Ce (cerium) 56.9% | Cl (chlorine) 43.1%
Structure diagrams

Structure diagrams
Basic properties
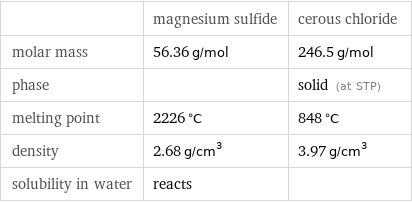
| magnesium sulfide | cerous chloride molar mass | 56.36 g/mol | 246.5 g/mol phase | | solid (at STP) melting point | 2226 °C | 848 °C density | 2.68 g/cm^3 | 3.97 g/cm^3 solubility in water | reacts |
Units
