Input interpretation

group 9 elements | atomic volume
Results

cobalt | 1.03×10^7 pm^3 (cubic picometers) rhodium | 1.03×10^7 pm^3 (cubic picometers) iridium | 1.03×10^7 pm^3 (cubic picometers) meitnerium | (data not available)
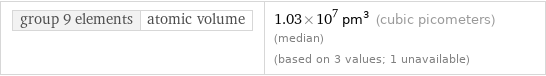
group 9 elements | atomic volume | 1.03×10^7 pm^3 (cubic picometers) (median) (based on 3 values; 1 unavailable)
Ranked values

| | visual | ratios | 3 | cobalt | | 1 | 1 2 | rhodium | | 1 | 1 1 | iridium | | 1 | 1
Plot
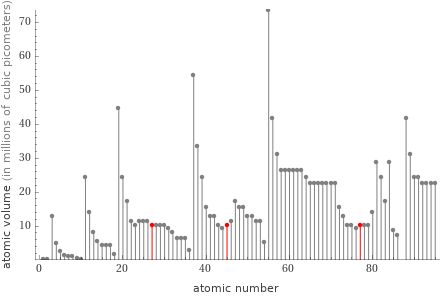
Plot
Periodic table location
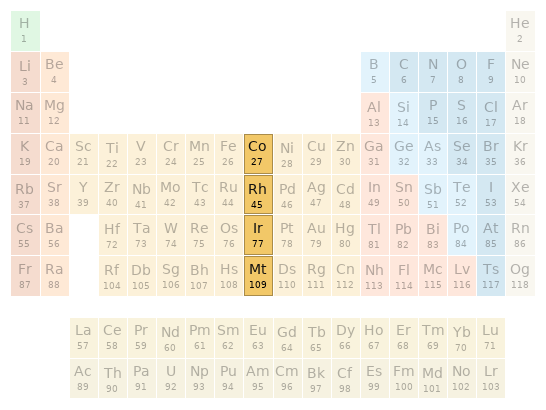
Periodic table location
Atomic radii properties

atomic radius (empirical) | median | 135 pm | highest | 135 pm (3 elements) | lowest | 135 pm (3 elements) atomic radius (calculated) | median | 173 pm | highest | 180 pm (iridium) | lowest | 152 pm (cobalt) | distribution | covalent radius | median | 141 pm | highest | 142 pm (rhodium) | lowest | 126 pm (cobalt)
Units

Unit conversions for median atomic volume 1.03×10^7 pm^3
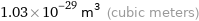
1.03×10^-29 m^3 (cubic meters)
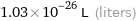
1.03×10^-26 L (liters)
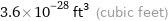
3.6×10^-28 ft^3 (cubic feet)
Comparison for median atomic volume 1.03×10^7 pm^3

≈ 83 × volume of a helium atom ( 4 Pi/3 atomic radii of helium (the smallest atom)^3 )
Corresponding quantities

Radius r of a sphere from V = 4πr^3/3: | 135 pm (picometers) | 5.3×10^-9 inches

Edge length a of a cube from V = a^3: | 218 pm (picometers) | 8.6×10^-9 inches