Input interpretation

monatomic anions | ionic conductivity
Summary

median | 76.56 cm^2/(Ω mol) highest | 78.1 cm^2/(Ω mol) (bromide anion) lowest | 55.4 cm^2/(Ω mol) (fluoride anion) | (based on 4 values; 8 unavailable)
Entities with missing values
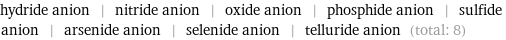
hydride anion | nitride anion | oxide anion | phosphide anion | sulfide anion | arsenide anion | selenide anion | telluride anion (total: 8)
Ionic conductivity rankings
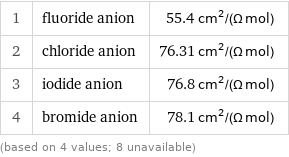
1 | fluoride anion | 55.4 cm^2/(Ω mol) 2 | chloride anion | 76.31 cm^2/(Ω mol) 3 | iodide anion | 76.8 cm^2/(Ω mol) 4 | bromide anion | 78.1 cm^2/(Ω mol) (based on 4 values; 8 unavailable)
Unit conversions for median ionic conductivity 76.56 cm^2/(Ω mol)

0.007656 m^2/(Ω mol) (meters squared per ohm mole)
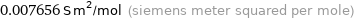
0.007656 S m^2/mol (siemens meter squared per mole)