Input interpretation
bromine iodine | atomic radius
Results
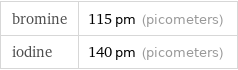
bromine | 115 pm (picometers) iodine | 140 pm (picometers)
Relative values
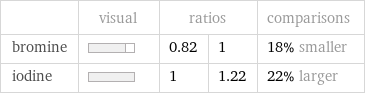
| visual | ratios | | comparisons bromine | | 0.82 | 1 | 18% smaller iodine | | 1 | 1.22 | 22% larger
Plot
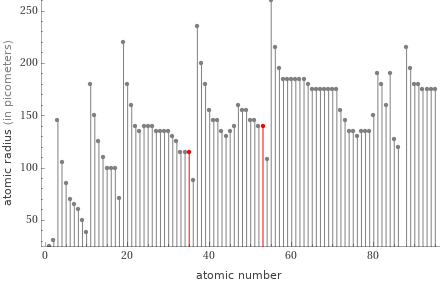
Plot
Periodic table location
Periodic table location
Atomic radii properties
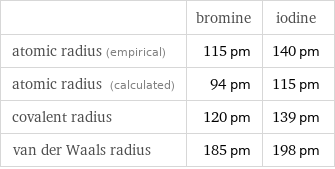
| bromine | iodine atomic radius (empirical) | 115 pm | 140 pm atomic radius (calculated) | 94 pm | 115 pm covalent radius | 120 pm | 139 pm van der Waals radius | 185 pm | 198 pm
Units
