Input interpretation
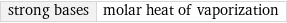
strong bases | molar heat of vaporization
Molar heat of vaporization rankings

1 | potassium hydroxide | 136 kJ/mol 2 | sodium hydroxide | 175 kJ/mol (based on 2 values; 13 unavailable)
Unit conversions for median molar heat of vaporization 155 kJ/mol
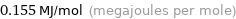
0.155 MJ/mol (megajoules per mole)
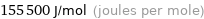
155500 J/mol (joules per mole)

37.2 kcal/mol (thermochemical kilocalories per mole)
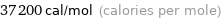
37200 cal/mol (calories per mole)
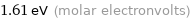
1.61 eV (molar electronvolts)