Input interpretation

naturally occurring elements | melting point
Summary

median | 931 °C highest | 3550 °C (carbon) lowest | -259.14 °C (hydrogen) distribution | | (based on 87 values; 2 unavailable)
Entities with missing values
francium | helium (total: 2)
Plot
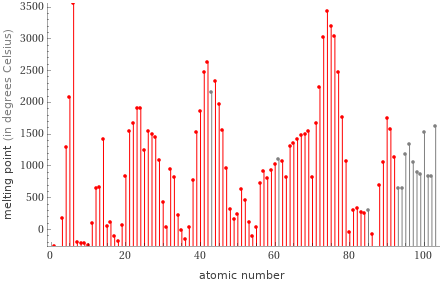
Plot
Distribution plots
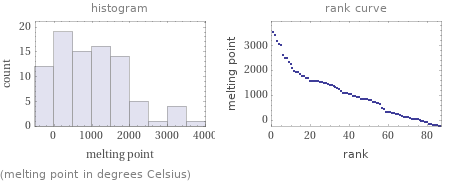
(melting point in degrees Celsius)
Melting point rankings
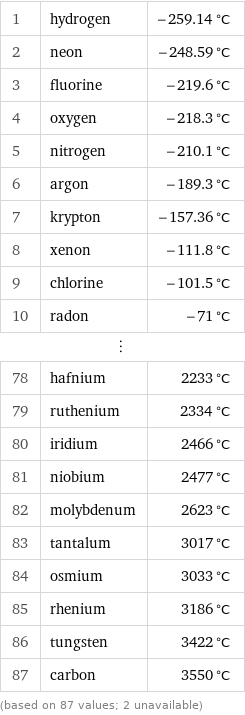
1 | hydrogen | -259.14 °C 2 | neon | -248.59 °C 3 | fluorine | -219.6 °C 4 | oxygen | -218.3 °C 5 | nitrogen | -210.1 °C 6 | argon | -189.3 °C 7 | krypton | -157.36 °C 8 | xenon | -111.8 °C 9 | chlorine | -101.5 °C 10 | radon | -71 °C ⋮ | | 78 | hafnium | 2233 °C 79 | ruthenium | 2334 °C 80 | iridium | 2466 °C 81 | niobium | 2477 °C 82 | molybdenum | 2623 °C 83 | tantalum | 3017 °C 84 | osmium | 3033 °C 85 | rhenium | 3186 °C 86 | tungsten | 3422 °C 87 | carbon | 3550 °C (based on 87 values; 2 unavailable)
Unit conversions for median melting point 931 °C
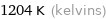
1204 K (kelvins)

1710 °F (degrees Fahrenheit)
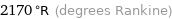
2170 °R (degrees Rankine)

745 °Ré (degrees Réaumur)
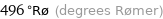
496 °Rø (degrees Rømer)
Comparison for median melting point 931 °C

369 °C below to 231 °C above typical temperature of magma (700 to 1300 °C)
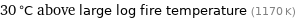
30 °C above large log fire temperature (1170 K)
Blackbody information for median melting point 931 °C (degrees Celsius)
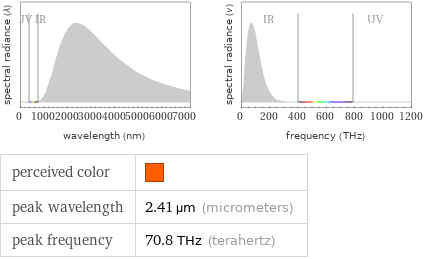
perceived color | peak wavelength | 2.41 µm (micrometers) peak frequency | 70.8 THz (terahertz)
Corresponding quantities
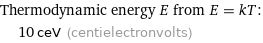
Thermodynamic energy E from E = kT: | 10 ceV (centielectronvolts)
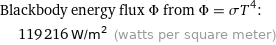
Blackbody energy flux Φ from Φ = σT^4: | 119216 W/m^2 (watts per square meter)
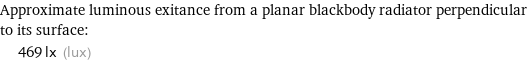
Approximate luminous exitance from a planar blackbody radiator perpendicular to its surface: | 469 lx (lux)
Thermodynamic properties
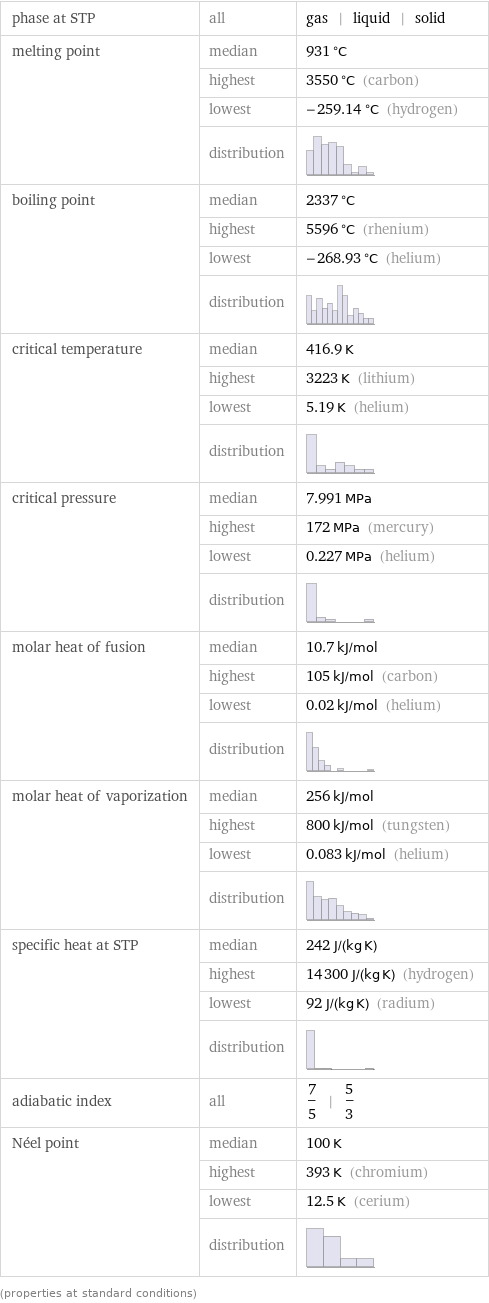
phase at STP | all | gas | liquid | solid melting point | median | 931 °C | highest | 3550 °C (carbon) | lowest | -259.14 °C (hydrogen) | distribution | boiling point | median | 2337 °C | highest | 5596 °C (rhenium) | lowest | -268.93 °C (helium) | distribution | critical temperature | median | 416.9 K | highest | 3223 K (lithium) | lowest | 5.19 K (helium) | distribution | critical pressure | median | 7.991 MPa | highest | 172 MPa (mercury) | lowest | 0.227 MPa (helium) | distribution | molar heat of fusion | median | 10.7 kJ/mol | highest | 105 kJ/mol (carbon) | lowest | 0.02 kJ/mol (helium) | distribution | molar heat of vaporization | median | 256 kJ/mol | highest | 800 kJ/mol (tungsten) | lowest | 0.083 kJ/mol (helium) | distribution | specific heat at STP | median | 242 J/(kg K) | highest | 14300 J/(kg K) (hydrogen) | lowest | 92 J/(kg K) (radium) | distribution | adiabatic index | all | 7/5 | 5/3 Néel point | median | 100 K | highest | 393 K (chromium) | lowest | 12.5 K (cerium) | distribution | (properties at standard conditions)