Input interpretation
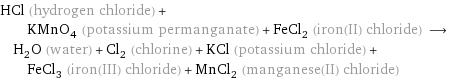
HCl (hydrogen chloride) + KMnO_4 (potassium permanganate) + FeCl_2 (iron(II) chloride) ⟶ H_2O (water) + Cl_2 (chlorine) + KCl (potassium chloride) + FeCl_3 (iron(III) chloride) + MnCl_2 (manganese(II) chloride)
Balanced equation

Balance the chemical equation algebraically: HCl + KMnO_4 + FeCl_2 ⟶ H_2O + Cl_2 + KCl + FeCl_3 + MnCl_2 Add stoichiometric coefficients, c_i, to the reactants and products: c_1 HCl + c_2 KMnO_4 + c_3 FeCl_2 ⟶ c_4 H_2O + c_5 Cl_2 + c_6 KCl + c_7 FeCl_3 + c_8 MnCl_2 Set the number of atoms in the reactants equal to the number of atoms in the products for Cl, H, K, Mn, O and Fe: Cl: | c_1 + 2 c_3 = 2 c_5 + c_6 + 3 c_7 + 2 c_8 H: | c_1 = 2 c_4 K: | c_2 = c_6 Mn: | c_2 = c_8 O: | 4 c_2 = c_4 Fe: | c_3 = c_7 Since the coefficients are relative quantities and underdetermined, choose a coefficient to set arbitrarily. To keep the coefficients small, the arbitrary value is ordinarily one. For instance, set c_2 = 1 and solve the system of equations for the remaining coefficients: c_1 = 8 c_2 = 1 c_4 = 4 c_5 = 5/2 - c_3/2 c_6 = 1 c_7 = c_3 c_8 = 1 The resulting system of equations is still underdetermined, so an additional coefficient must be set arbitrarily. Set c_3 = 1 and solve for the remaining coefficients: c_1 = 8 c_2 = 1 c_3 = 1 c_4 = 4 c_5 = 2 c_6 = 1 c_7 = 1 c_8 = 1 Substitute the coefficients into the chemical reaction to obtain the balanced equation: Answer: | | 8 HCl + KMnO_4 + FeCl_2 ⟶ 4 H_2O + 2 Cl_2 + KCl + FeCl_3 + MnCl_2
Structures
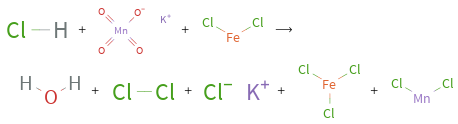
+ + ⟶ + + + +
Names
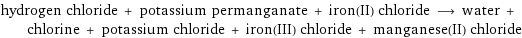
hydrogen chloride + potassium permanganate + iron(II) chloride ⟶ water + chlorine + potassium chloride + iron(III) chloride + manganese(II) chloride
Reaction thermodynamics
Gibbs free energy
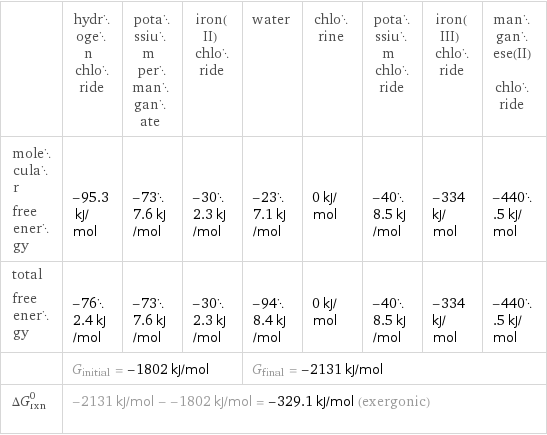
| hydrogen chloride | potassium permanganate | iron(II) chloride | water | chlorine | potassium chloride | iron(III) chloride | manganese(II) chloride molecular free energy | -95.3 kJ/mol | -737.6 kJ/mol | -302.3 kJ/mol | -237.1 kJ/mol | 0 kJ/mol | -408.5 kJ/mol | -334 kJ/mol | -440.5 kJ/mol total free energy | -762.4 kJ/mol | -737.6 kJ/mol | -302.3 kJ/mol | -948.4 kJ/mol | 0 kJ/mol | -408.5 kJ/mol | -334 kJ/mol | -440.5 kJ/mol | G_initial = -1802 kJ/mol | | | G_final = -2131 kJ/mol | | | | ΔG_rxn^0 | -2131 kJ/mol - -1802 kJ/mol = -329.1 kJ/mol (exergonic) | | | | | | |
Equilibrium constant
![K_c = ([H2O]^4 [Cl2]^2 [KCl] [FeCl3] [MnCl2])/([HCl]^8 [KMnO4] [FeCl2])](../image_source/865fcdfdf6e88afeeb5f72a9ba8f0573.png)
K_c = ([H2O]^4 [Cl2]^2 [KCl] [FeCl3] [MnCl2])/([HCl]^8 [KMnO4] [FeCl2])
Rate of reaction
![rate = -1/8 (Δ[HCl])/(Δt) = -(Δ[KMnO4])/(Δt) = -(Δ[FeCl2])/(Δt) = 1/4 (Δ[H2O])/(Δt) = 1/2 (Δ[Cl2])/(Δt) = (Δ[KCl])/(Δt) = (Δ[FeCl3])/(Δt) = (Δ[MnCl2])/(Δt) (assuming constant volume and no accumulation of intermediates or side products)](../image_source/e1c049f1e851e35765971e054df29620.png)
rate = -1/8 (Δ[HCl])/(Δt) = -(Δ[KMnO4])/(Δt) = -(Δ[FeCl2])/(Δt) = 1/4 (Δ[H2O])/(Δt) = 1/2 (Δ[Cl2])/(Δt) = (Δ[KCl])/(Δt) = (Δ[FeCl3])/(Δt) = (Δ[MnCl2])/(Δt) (assuming constant volume and no accumulation of intermediates or side products)
Chemical names and formulas
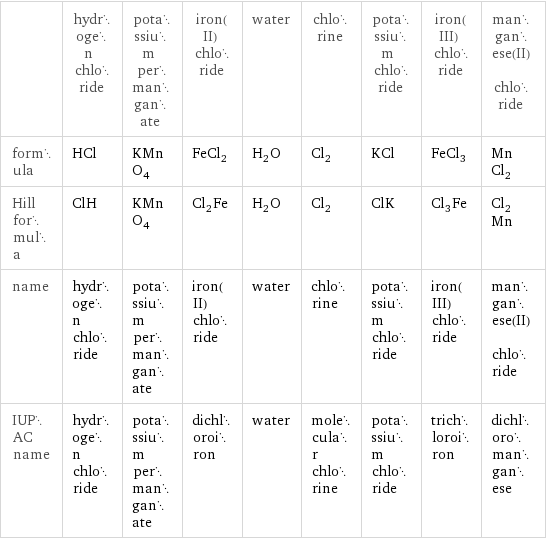
| hydrogen chloride | potassium permanganate | iron(II) chloride | water | chlorine | potassium chloride | iron(III) chloride | manganese(II) chloride formula | HCl | KMnO_4 | FeCl_2 | H_2O | Cl_2 | KCl | FeCl_3 | MnCl_2 Hill formula | ClH | KMnO_4 | Cl_2Fe | H_2O | Cl_2 | ClK | Cl_3Fe | Cl_2Mn name | hydrogen chloride | potassium permanganate | iron(II) chloride | water | chlorine | potassium chloride | iron(III) chloride | manganese(II) chloride IUPAC name | hydrogen chloride | potassium permanganate | dichloroiron | water | molecular chlorine | potassium chloride | trichloroiron | dichloromanganese