Input interpretation
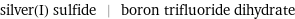
silver(I) sulfide | boron trifluoride dihydrate
Chemical names and formulas

| silver(I) sulfide | boron trifluoride dihydrate formula | Ag_2S | BF_3·2H_2O Hill formula | Ag_2S_1 | BF_3H_4O_2 name | silver(I) sulfide | boron trifluoride dihydrate IUPAC name | | trifluoroborane dihydrate alternate names | silver; silver; sulfanide | silver sulfide | trifluoroborane dihydrate mass fractions | Ag (silver) 77.1% | S (sulfur) 22.9% | B (boron) 10.4% | F (fluorine) 54.9% | H (hydrogen) 3.88% | O (oxygen) 30.8%
Structure diagrams
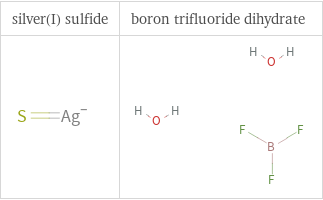
Structure diagrams
Basic properties
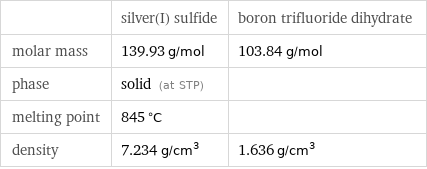
| silver(I) sulfide | boron trifluoride dihydrate molar mass | 139.93 g/mol | 103.84 g/mol phase | solid (at STP) | melting point | 845 °C | density | 7.234 g/cm^3 | 1.636 g/cm^3
Units

Solid properties
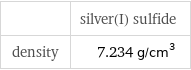
| silver(I) sulfide density | 7.234 g/cm^3
Units
