Input interpretation
astatine (chemical element) | iodine (chemical element) | argon (chemical element)
Periodic table location
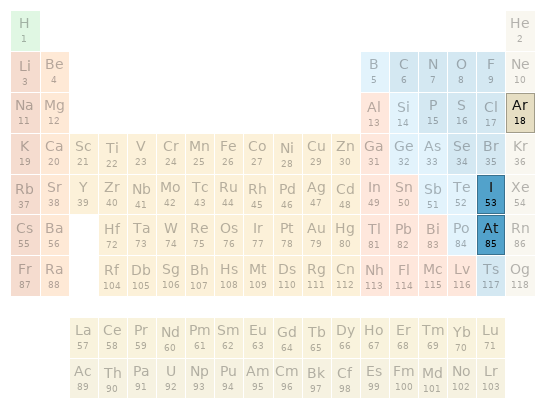
Periodic table location
Images

Images
Basic elemental properties
![| astatine | iodine | argon atomic symbol | At | I | Ar atomic number | 85 | 53 | 18 short electronic configuration | [Xe]6s^24f^145d^106p^5 | [Kr]5s^24d^105p^5 | [Ne]3s^23p^6 Aufbau diagram | 6p 5d 4f 6s | 5p 4d 5s | 3p 3s block | p | p | p group | 17 | 17 | 18 period | 6 | 5 | 3 atomic mass | 210 u | 126.90447 u | 39.948 u half-life | 8.1 h | (stable) | (stable)](../image_source/e1c1958db6a033ed17280e64e0413256.png)
| astatine | iodine | argon atomic symbol | At | I | Ar atomic number | 85 | 53 | 18 short electronic configuration | [Xe]6s^24f^145d^106p^5 | [Kr]5s^24d^105p^5 | [Ne]3s^23p^6 Aufbau diagram | 6p 5d 4f 6s | 5p 4d 5s | 3p 3s block | p | p | p group | 17 | 17 | 18 period | 6 | 5 | 3 atomic mass | 210 u | 126.90447 u | 39.948 u half-life | 8.1 h | (stable) | (stable)
Thermodynamic properties
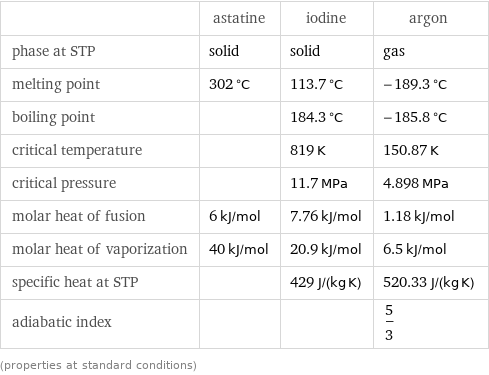
| astatine | iodine | argon phase at STP | solid | solid | gas melting point | 302 °C | 113.7 °C | -189.3 °C boiling point | | 184.3 °C | -185.8 °C critical temperature | | 819 K | 150.87 K critical pressure | | 11.7 MPa | 4.898 MPa molar heat of fusion | 6 kJ/mol | 7.76 kJ/mol | 1.18 kJ/mol molar heat of vaporization | 40 kJ/mol | 20.9 kJ/mol | 6.5 kJ/mol specific heat at STP | | 429 J/(kg K) | 520.33 J/(kg K) adiabatic index | | | 5/3 (properties at standard conditions)
Material properties
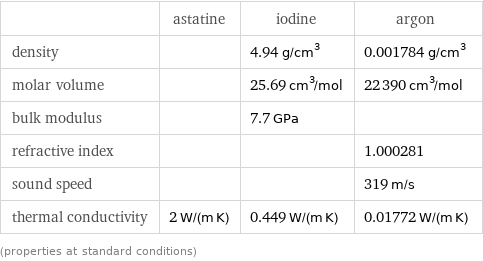
| astatine | iodine | argon density | | 4.94 g/cm^3 | 0.001784 g/cm^3 molar volume | | 25.69 cm^3/mol | 22390 cm^3/mol bulk modulus | | 7.7 GPa | refractive index | | | 1.000281 sound speed | | | 319 m/s thermal conductivity | 2 W/(m K) | 0.449 W/(m K) | 0.01772 W/(m K) (properties at standard conditions)
Electromagnetic properties
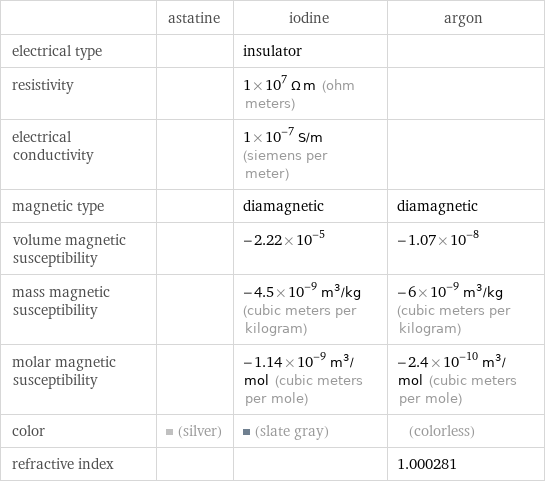
| astatine | iodine | argon electrical type | | insulator | resistivity | | 1×10^7 Ω m (ohm meters) | electrical conductivity | | 1×10^-7 S/m (siemens per meter) | magnetic type | | diamagnetic | diamagnetic volume magnetic susceptibility | | -2.22×10^-5 | -1.07×10^-8 mass magnetic susceptibility | | -4.5×10^-9 m^3/kg (cubic meters per kilogram) | -6×10^-9 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | | -1.14×10^-9 m^3/mol (cubic meters per mole) | -2.4×10^-10 m^3/mol (cubic meters per mole) color | (silver) | (slate gray) | (colorless) refractive index | | | 1.000281
Reactivity
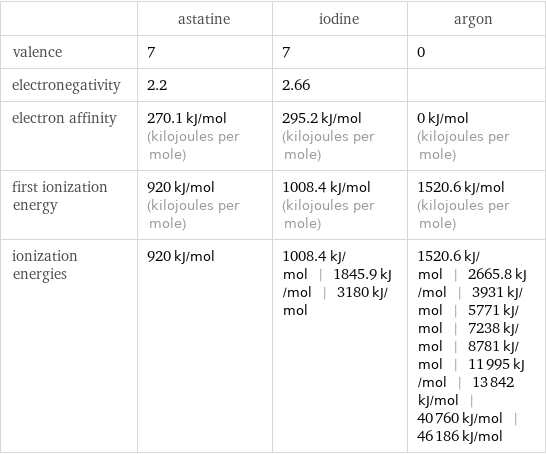
| astatine | iodine | argon valence | 7 | 7 | 0 electronegativity | 2.2 | 2.66 | electron affinity | 270.1 kJ/mol (kilojoules per mole) | 295.2 kJ/mol (kilojoules per mole) | 0 kJ/mol (kilojoules per mole) first ionization energy | 920 kJ/mol (kilojoules per mole) | 1008.4 kJ/mol (kilojoules per mole) | 1520.6 kJ/mol (kilojoules per mole) ionization energies | 920 kJ/mol | 1008.4 kJ/mol | 1845.9 kJ/mol | 3180 kJ/mol | 1520.6 kJ/mol | 2665.8 kJ/mol | 3931 kJ/mol | 5771 kJ/mol | 7238 kJ/mol | 8781 kJ/mol | 11995 kJ/mol | 13842 kJ/mol | 40760 kJ/mol | 46186 kJ/mol
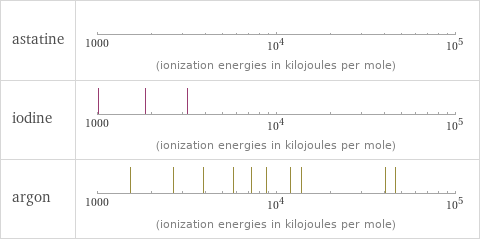
Reactivity
Atomic properties

| astatine | iodine | argon term symbol | ^2P_(3/2) | ^2P_(3/2) | ^1S_0 atomic radius | 127 pm | 140 pm | 71 pm covalent radius | 150 pm | 139 pm | 106 pm van der Waals radius | | 198 pm | 188 pm (electronic ground state properties)
Abundances
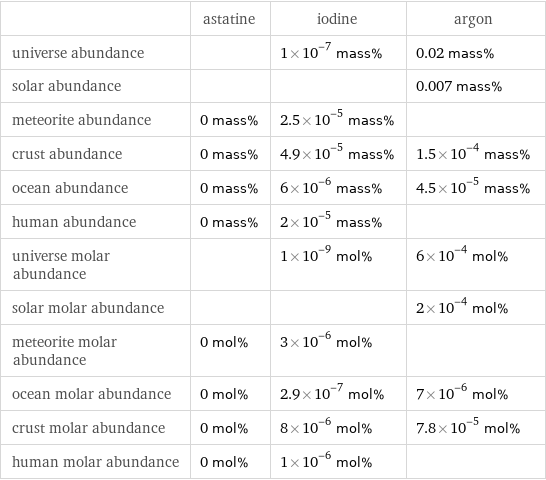
| astatine | iodine | argon universe abundance | | 1×10^-7 mass% | 0.02 mass% solar abundance | | | 0.007 mass% meteorite abundance | 0 mass% | 2.5×10^-5 mass% | crust abundance | 0 mass% | 4.9×10^-5 mass% | 1.5×10^-4 mass% ocean abundance | 0 mass% | 6×10^-6 mass% | 4.5×10^-5 mass% human abundance | 0 mass% | 2×10^-5 mass% | universe molar abundance | | 1×10^-9 mol% | 6×10^-4 mol% solar molar abundance | | | 2×10^-4 mol% meteorite molar abundance | 0 mol% | 3×10^-6 mol% | ocean molar abundance | 0 mol% | 2.9×10^-7 mol% | 7×10^-6 mol% crust molar abundance | 0 mol% | 8×10^-6 mol% | 7.8×10^-5 mol% human molar abundance | 0 mol% | 1×10^-6 mol% |
Nuclear properties

| astatine | iodine | argon half-life | 8.1 h | (stable) | (stable) specific radioactivity | 68278 TBq/g | | decay mode | beta plus decay | | stable isotopes | | I-127 (100%) | Ar-40 (99.6003%) | Ar-36 (0.3365%) | Ar-38 (6.32×10^-4) nuclear spin | At-193: 1/2^+ | At-194: 3^+ | At-195: 1/2^+ | At-196: 3^+ | At-197: 9/2^- | At-198: 3^+ | At-199: 9/2^- | At-200: 3^+ | At-201: 9/2^- | At-203: 9/2^- | At-204: 7^+ | At-205: 9/2^- | At-206: 5^+ | At-207: 9/2^- | At-208: 6^+ | At-209: 9/2^- | At-210: 5^+ | At-211: 9/2^- | At-212: 1^- | At-213: 9/2^- | At-214: 1^- | At-215: 9/2^- | At-216: 1^- | At-217: 9/2^- | At-218: 1^- | At-219: 5/2^- | At-220: 3^- | At-221: 3/2^- | At-223: 3/2^- | I-108: 1^+ | I-109: 1/2^+ | I-110: 1^+ | I-111: 5/2^+ | I-112: 1^+ | I-113: 5/2^+ | I-114: 1^+ | I-115: 5/2^+ | I-116: 1^+ | I-117: 5/2^+ | I-118: 2^- | I-119: 5/2^+ | I-120: 2^- | I-121: 5/2^+ | I-122: 1^+ | I-123: 5/2^+ | I-124: 2^- | I-125: 5/2^+ | I-126: 2^- | I-127: 5/2^+ | I-128: 1^+ | I-129: 7/2^+ | I-130: 5^+ | I-131: 7/2^+ | I-132: 4^+ | I-133: 7/2^+ | I-134: 4^+ | I-135: 7/2^+ | I-136: 1^- | I-137: 7/2^+ | I-138: 2^- | I-139: 7/2^+ | I-140: 4^- | I-141: 7/2^+ | I-142: 2^- | I-143: 7/2^+ | I-144: 1^- | Ar-30: 0^+ | Ar-31: 5/2^+ | Ar-32: 0^+ | Ar-33: 1/2^+ | Ar-34: 0^+ | Ar-35: 3/2^+ | Ar-36: 0^+ | Ar-37: 3/2^+ | Ar-38: 0^+ | Ar-39: 7/2^- | Ar-40: 0^+ | Ar-41: 7/2^- | Ar-42: 0^+ | Ar-43: 5/2^- | Ar-44: 0^+ | Ar-46: 0^+ | Ar-47: 3/2^- | Ar-48: 0^+ | Ar-49: 3/2^- | Ar-50: 0^+ | Ar-51: 3/2^- | Ar-52: 0^+ | Ar-53: 5/2^- unstable isotopes | At-210 (8.1 h) | At-211 (7.2139 h) | At-209 (5.42 h) | At-207 (110 min) | At-208 (97.83 min) | At-206 (30.67 min) | At-205 (26.9 min) | At-204 (9.17 min) | At-203 (7.33 min) | At-220 (223 s) | At-202 (184 s) | At-221 (140 s) | At-201 (85 s) | At-219 (56 s) | At-222 (54 s) | At-223 (50 s) | At-200 (43.2 s) | At-199 (7.03 s) | At-198 (4.2 s) | At-218 (1.5 s) | At-197 (388 ms) | At-196 (388 ms) | At-195 (328 ms) | At-212 (314 ms) | At-194 (40 ms) | At-217 (32.3 ms) | At-193 (28 ms) | At-216 (300 µs) | At-215 (100 µs) | At-214 (558 ns) | At-213 (125 ns) | I-129 (15.7 Myr) | I-125 (59.4 days) | I-126 (12.93 days) | I-131 (8.0252 days) | I-124 (100.2 h) | I-133 (20.8 h) | I-123 (13.2235 h) | I-130 (12.4 h) | I-135 (6.58 h) | I-132 (137.7 min) | I-121 (127.2 min) | I-120 (82 min) | I-134 (52.5 min) | I-128 (24.98 min) | I-119 (19.1 min) | I-118 (13.7 min) | I-122 (218 s) | I-117 (133 s) | I-136 (83.4 s) | I-115 (78 s) | I-137 (24.5 s) | I-113 (6.6 s) | I-138 (6.23 s) | I-112 (3.42 s) | I-116 (2.91 s) | I-111 (2.5 s) | I-139 (2.28 s) | I-114 (2.1 s) | I-140 (860 ms) | I-110 (650 ms) | I-141 (430 ms) | I-142 (200 ms) | I-108 (36 ms) | I-109 (103 µs) | I-144 (300 ns) | I-143 (150 ns) | Ar-39 (269 yr) | Ar-42 (33 yr) | Ar-37 (34.95 days) | Ar-41 (109.61 min) | Ar-44 (11.87 min) | Ar-43 (5.37 min) | Ar-45 (21.48 s) | Ar-46 (8.4 s) | Ar-35 (1.775 s) | Ar-47 (1.23 s) | Ar-34 (845 ms) | Ar-48 (475 ms) | Ar-33 (173 ms) | Ar-49 (170 ms) | Ar-32 (98 ms) | Ar-31 (14.4 ms) | Ar-52 (10 ms) | Ar-53 (3 ms) | Ar-51 (200 ns) | Ar-50 (170 ns) | Ar-30 (20 ns) neutron cross-section | | 6.2 b | 0.65 b neutron mass absorption | | 0.0018 m^2/kg | 6×10^-4 m^2/kg
Identifiers
| astatine | iodine | argon CAS number | 7440-68-8 | 7553-56-2 | 7440-37-1 PubChem CID number | 5460479 | 807 | 23968