Input interpretation
arsenic (chemical element) | antimony (chemical element) | technetium (chemical element)
Periodic table location
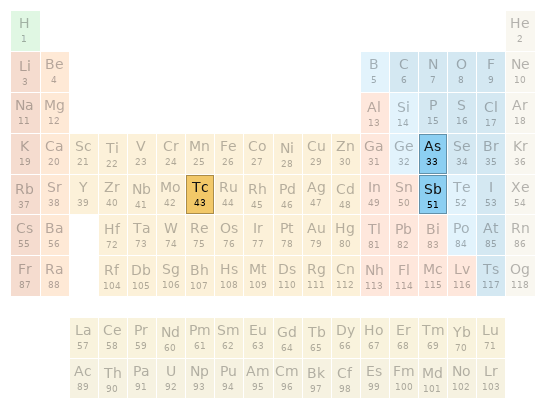
Periodic table location
Images

Images
Basic elemental properties
![| arsenic | antimony | technetium atomic symbol | As | Sb | Tc atomic number | 33 | 51 | 43 short electronic configuration | [Ar]4s^23d^104p^3 | [Kr]5s^24d^105p^3 | [Kr]5s^24d^5 Aufbau diagram | 4p 3d 4s | 5p 4d 5s | 4d 5s block | p | p | d group | 15 | 15 | 7 period | 4 | 5 | 5 atomic mass | 74.921595 u | 121.76 u | 98 u half-life | (stable) | (stable) | 0.21 Myr](../image_source/19b5798fb12ecabb1a95c72eda0266e9.png)
| arsenic | antimony | technetium atomic symbol | As | Sb | Tc atomic number | 33 | 51 | 43 short electronic configuration | [Ar]4s^23d^104p^3 | [Kr]5s^24d^105p^3 | [Kr]5s^24d^5 Aufbau diagram | 4p 3d 4s | 5p 4d 5s | 4d 5s block | p | p | d group | 15 | 15 | 7 period | 4 | 5 | 5 atomic mass | 74.921595 u | 121.76 u | 98 u half-life | (stable) | (stable) | 0.21 Myr
Thermodynamic properties
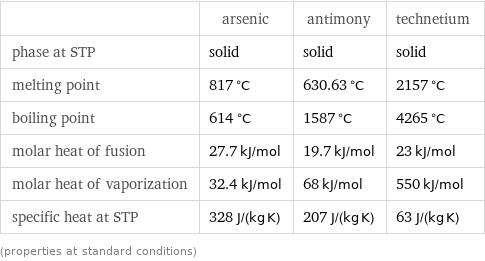
| arsenic | antimony | technetium phase at STP | solid | solid | solid melting point | 817 °C | 630.63 °C | 2157 °C boiling point | 614 °C | 1587 °C | 4265 °C molar heat of fusion | 27.7 kJ/mol | 19.7 kJ/mol | 23 kJ/mol molar heat of vaporization | 32.4 kJ/mol | 68 kJ/mol | 550 kJ/mol specific heat at STP | 328 J/(kg K) | 207 J/(kg K) | 63 J/(kg K) (properties at standard conditions)
Material properties
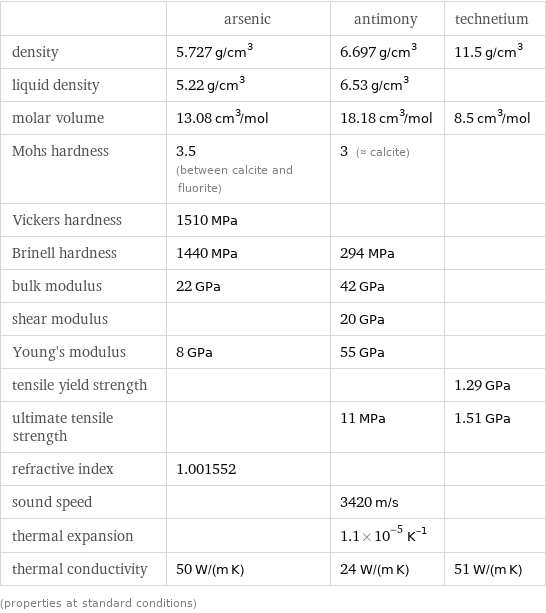
| arsenic | antimony | technetium density | 5.727 g/cm^3 | 6.697 g/cm^3 | 11.5 g/cm^3 liquid density | 5.22 g/cm^3 | 6.53 g/cm^3 | molar volume | 13.08 cm^3/mol | 18.18 cm^3/mol | 8.5 cm^3/mol Mohs hardness | 3.5 (between calcite and fluorite) | 3 (≈ calcite) | Vickers hardness | 1510 MPa | | Brinell hardness | 1440 MPa | 294 MPa | bulk modulus | 22 GPa | 42 GPa | shear modulus | | 20 GPa | Young's modulus | 8 GPa | 55 GPa | tensile yield strength | | | 1.29 GPa ultimate tensile strength | | 11 MPa | 1.51 GPa refractive index | 1.001552 | | sound speed | | 3420 m/s | thermal expansion | | 1.1×10^-5 K^(-1) | thermal conductivity | 50 W/(m K) | 24 W/(m K) | 51 W/(m K) (properties at standard conditions)
Electromagnetic properties
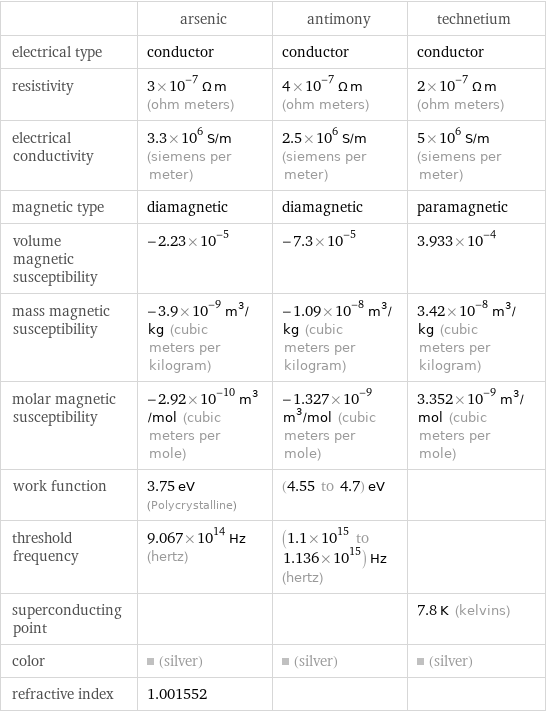
| arsenic | antimony | technetium electrical type | conductor | conductor | conductor resistivity | 3×10^-7 Ω m (ohm meters) | 4×10^-7 Ω m (ohm meters) | 2×10^-7 Ω m (ohm meters) electrical conductivity | 3.3×10^6 S/m (siemens per meter) | 2.5×10^6 S/m (siemens per meter) | 5×10^6 S/m (siemens per meter) magnetic type | diamagnetic | diamagnetic | paramagnetic volume magnetic susceptibility | -2.23×10^-5 | -7.3×10^-5 | 3.933×10^-4 mass magnetic susceptibility | -3.9×10^-9 m^3/kg (cubic meters per kilogram) | -1.09×10^-8 m^3/kg (cubic meters per kilogram) | 3.42×10^-8 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | -2.92×10^-10 m^3/mol (cubic meters per mole) | -1.327×10^-9 m^3/mol (cubic meters per mole) | 3.352×10^-9 m^3/mol (cubic meters per mole) work function | 3.75 eV (Polycrystalline) | (4.55 to 4.7) eV | threshold frequency | 9.067×10^14 Hz (hertz) | (1.1×10^15 to 1.136×10^15) Hz (hertz) | superconducting point | | | 7.8 K (kelvins) color | (silver) | (silver) | (silver) refractive index | 1.001552 | |
Reactivity
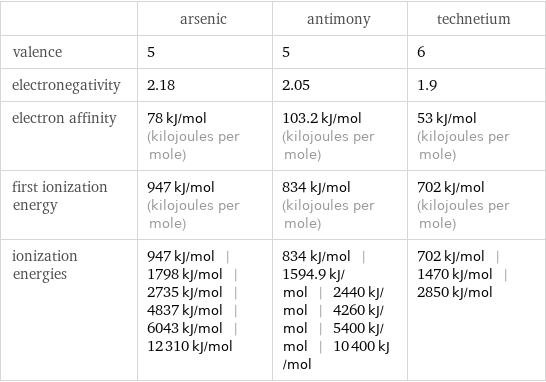
| arsenic | antimony | technetium valence | 5 | 5 | 6 electronegativity | 2.18 | 2.05 | 1.9 electron affinity | 78 kJ/mol (kilojoules per mole) | 103.2 kJ/mol (kilojoules per mole) | 53 kJ/mol (kilojoules per mole) first ionization energy | 947 kJ/mol (kilojoules per mole) | 834 kJ/mol (kilojoules per mole) | 702 kJ/mol (kilojoules per mole) ionization energies | 947 kJ/mol | 1798 kJ/mol | 2735 kJ/mol | 4837 kJ/mol | 6043 kJ/mol | 12310 kJ/mol | 834 kJ/mol | 1594.9 kJ/mol | 2440 kJ/mol | 4260 kJ/mol | 5400 kJ/mol | 10400 kJ/mol | 702 kJ/mol | 1470 kJ/mol | 2850 kJ/mol
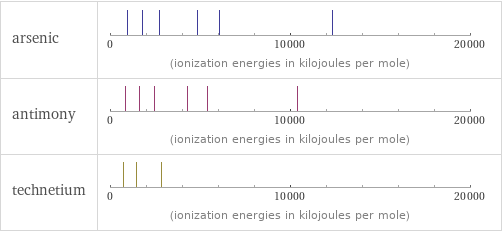
Reactivity
Atomic properties
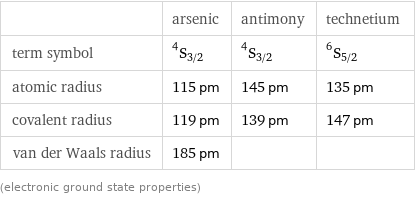
| arsenic | antimony | technetium term symbol | ^4S_(3/2) | ^4S_(3/2) | ^6S_(5/2) atomic radius | 115 pm | 145 pm | 135 pm covalent radius | 119 pm | 139 pm | 147 pm van der Waals radius | 185 pm | | (electronic ground state properties)
Abundances
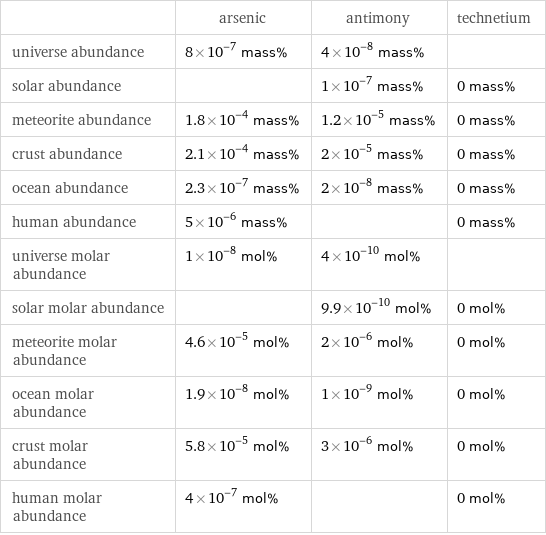
| arsenic | antimony | technetium universe abundance | 8×10^-7 mass% | 4×10^-8 mass% | solar abundance | | 1×10^-7 mass% | 0 mass% meteorite abundance | 1.8×10^-4 mass% | 1.2×10^-5 mass% | 0 mass% crust abundance | 2.1×10^-4 mass% | 2×10^-5 mass% | 0 mass% ocean abundance | 2.3×10^-7 mass% | 2×10^-8 mass% | 0 mass% human abundance | 5×10^-6 mass% | | 0 mass% universe molar abundance | 1×10^-8 mol% | 4×10^-10 mol% | solar molar abundance | | 9.9×10^-10 mol% | 0 mol% meteorite molar abundance | 4.6×10^-5 mol% | 2×10^-6 mol% | 0 mol% ocean molar abundance | 1.9×10^-8 mol% | 1×10^-9 mol% | 0 mol% crust molar abundance | 5.8×10^-5 mol% | 3×10^-6 mol% | 0 mol% human molar abundance | 4×10^-7 mol% | | 0 mol%