Input interpretation
zinc chlorate | magnesium nitrate
Chemical names and formulas
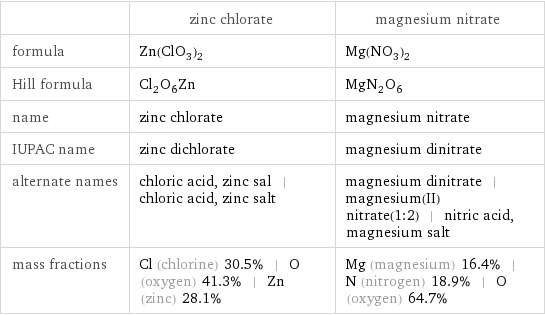
| zinc chlorate | magnesium nitrate formula | Zn(ClO_3)_2 | Mg(NO_3)_2 Hill formula | Cl_2O_6Zn | MgN_2O_6 name | zinc chlorate | magnesium nitrate IUPAC name | zinc dichlorate | magnesium dinitrate alternate names | chloric acid, zinc sal | chloric acid, zinc salt | magnesium dinitrate | magnesium(II) nitrate(1:2) | nitric acid, magnesium salt mass fractions | Cl (chlorine) 30.5% | O (oxygen) 41.3% | Zn (zinc) 28.1% | Mg (magnesium) 16.4% | N (nitrogen) 18.9% | O (oxygen) 64.7%
Structure diagrams
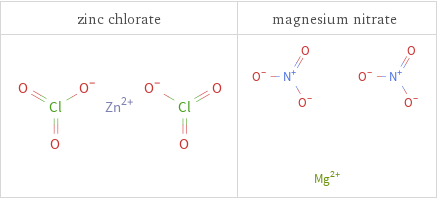
Structure diagrams
Basic properties
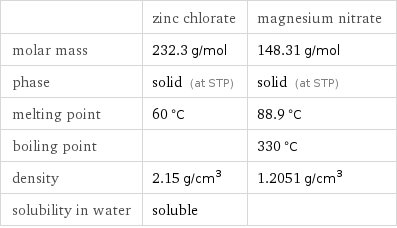
| zinc chlorate | magnesium nitrate molar mass | 232.3 g/mol | 148.31 g/mol phase | solid (at STP) | solid (at STP) melting point | 60 °C | 88.9 °C boiling point | | 330 °C density | 2.15 g/cm^3 | 1.2051 g/cm^3 solubility in water | soluble |
Units
