Input interpretation

period 2 elements | most common oxidation states
Table
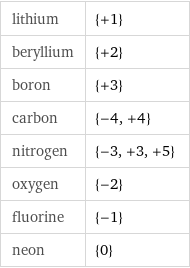
lithium | {+1} beryllium | {+2} boron | {+3} carbon | {-4, +4} nitrogen | {-3, +3, +5} oxygen | {-2} fluorine | {-1} neon | {0}