Input interpretation
elements | phase at STP
Summary
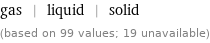
gas | liquid | solid (based on 99 values; 19 unavailable)
Entities with missing values
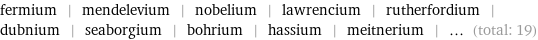
fermium | mendelevium | nobelium | lawrencium | rutherfordium | dubnium | seaborgium | bohrium | hassium | meitnerium | ... (total: 19)
Categorized list

gas | argon, chlorine, fluorine, helium, hydrogen, krypton, neon, nitrogen, oxygen, radon, xenon liquid | bromine, mercury solid | actinium, aluminum, americium, antimony, arsenic, astatine, barium, berkelium, beryllium, bismuth, boron, cadmium, calcium, californium, carbon, cerium, cesium, chromium, cobalt, copper, ...
Thermodynamic properties
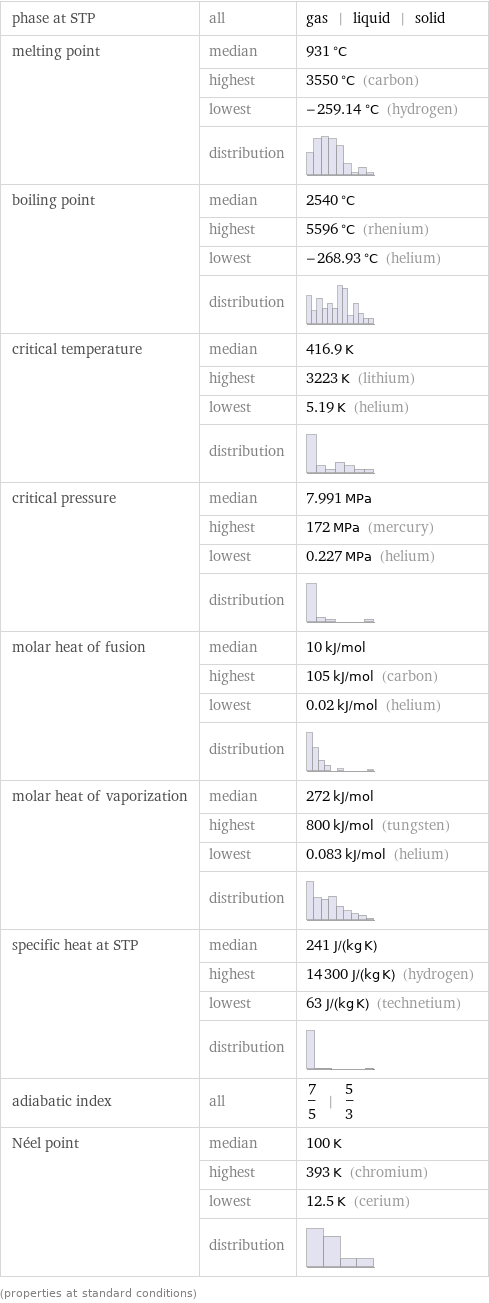
phase at STP | all | gas | liquid | solid melting point | median | 931 °C | highest | 3550 °C (carbon) | lowest | -259.14 °C (hydrogen) | distribution | boiling point | median | 2540 °C | highest | 5596 °C (rhenium) | lowest | -268.93 °C (helium) | distribution | critical temperature | median | 416.9 K | highest | 3223 K (lithium) | lowest | 5.19 K (helium) | distribution | critical pressure | median | 7.991 MPa | highest | 172 MPa (mercury) | lowest | 0.227 MPa (helium) | distribution | molar heat of fusion | median | 10 kJ/mol | highest | 105 kJ/mol (carbon) | lowest | 0.02 kJ/mol (helium) | distribution | molar heat of vaporization | median | 272 kJ/mol | highest | 800 kJ/mol (tungsten) | lowest | 0.083 kJ/mol (helium) | distribution | specific heat at STP | median | 241 J/(kg K) | highest | 14300 J/(kg K) (hydrogen) | lowest | 63 J/(kg K) (technetium) | distribution | adiabatic index | all | 7/5 | 5/3 Néel point | median | 100 K | highest | 393 K (chromium) | lowest | 12.5 K (cerium) | distribution | (properties at standard conditions)