Input interpretation
calcium nitrate
Chemical names and formulas

formula | Ca(NO_3)_2 Hill formula | CaN_2O_6 name | calcium nitrate IUPAC name | calcium dinitrate alternate names | calcium dinitrate | lime nitrate mass fractions | Ca (calcium) 24.4% | N (nitrogen) 17.1% | O (oxygen) 58.5%
Structure diagram
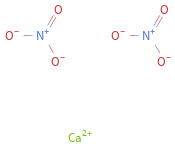
Structure diagram
Basic properties
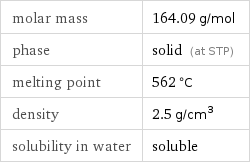
molar mass | 164.09 g/mol phase | solid (at STP) melting point | 562 °C density | 2.5 g/cm^3 solubility in water | soluble
Units

Solid properties (at STP)

density | 2.5 g/cm^3
Units

Thermodynamic properties
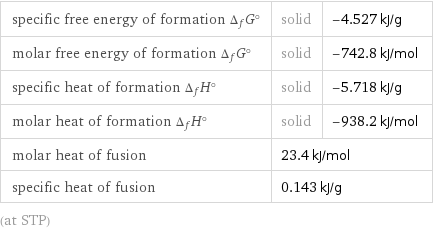
specific free energy of formation Δ_fG° | solid | -4.527 kJ/g molar free energy of formation Δ_fG° | solid | -742.8 kJ/mol specific heat of formation Δ_fH° | solid | -5.718 kJ/g molar heat of formation Δ_fH° | solid | -938.2 kJ/mol molar heat of fusion | 23.4 kJ/mol | specific heat of fusion | 0.143 kJ/g | (at STP)
Chemical identifiers
([O-])[O-].[N+](=O)([O-])[O-].[Ca+2] InChI identifier | InChI=1/Ca.2NO3/c;2*2-1(3)4/q+2;2*-1 EU number | 233-332-1 Gmelin number | 6516 RTECS number | EW2985000](../image_source/9ab0ca3de16afb92d363ddd0bf6ef981.png)
CAS number | 10124-37-5 PubChem CID number | 24963 SMILES identifier | [N+](=O)([O-])[O-].[N+](=O)([O-])[O-].[Ca+2] InChI identifier | InChI=1/Ca.2NO3/c;2*2-1(3)4/q+2;2*-1 EU number | 233-332-1 Gmelin number | 6516 RTECS number | EW2985000
NFPA label
NFPA label
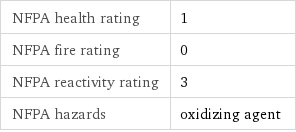
NFPA health rating | 1 NFPA fire rating | 0 NFPA reactivity rating | 3 NFPA hazards | oxidizing agent
Toxicity properties

RTECS classes | other
Ion equivalents

Ca^(2+) (calcium cation) | 1 (NO_3)^- (nitrate anion) | 2